Neurogenesis or non neurogenesis: that is the question
Full text
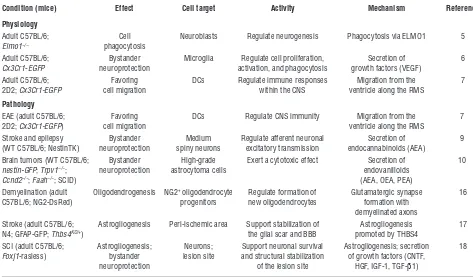
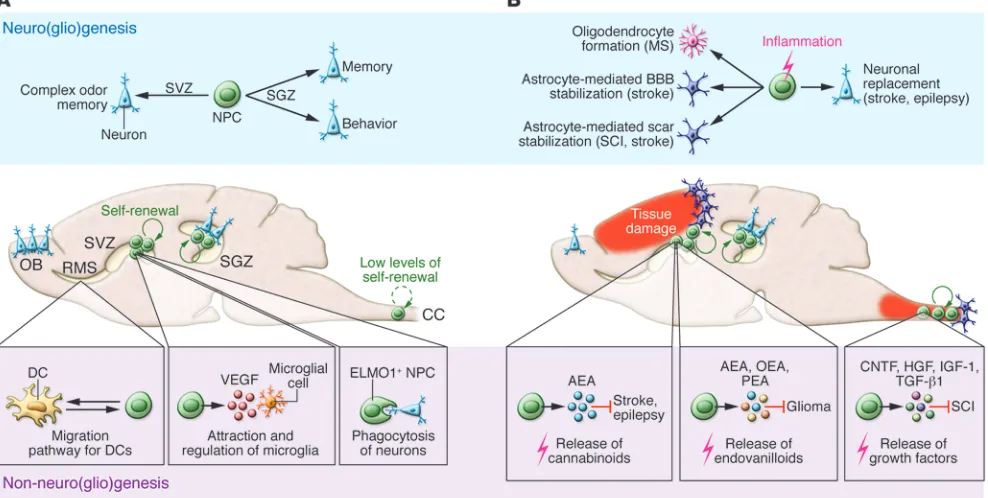
Figure
Related documents
savings finance emigration and worker remittances serve to make staying rather than migrating possible until a certain value, beyond which the opposite holds; (ii) lagged
We have estimated equations for migration, GDP per capita growth, labour force growth, remittances, aid, savings, investment, interest rates, literacy, tax revenues and
.1 An event record may be achieved by any athlete member of USA-S Swimming or his/her nation’s FINA-member organization who swims the time at a competition sanctioned or approved
This group consisted of five parents of youth with diabetes (ages 11–21 years). Four were female. Three of their children were diagnosed with type 2 diabetes. At the camp,
PUBLIC GAMING PER ONE INHABITANT 2007-2010 GAMING REVENUE BY MAIN GAME TYPES 2007-2010 NUMBER OF GAMES BY MAIN GAME TYPES 2007-2010 PRIZES 2007-2010.. SHARE OF EACH GAME FROM
20 The tax system in Philadelphia places its school district at such a disadvantage that without the state providing additional assistance, the relatively high revenue from