Blood
Transfusion
Effect
on the Respiratory
Pattern
of Preterm
Infants
Ashok
Joshi,
MD, Tilo Gerhardt,
MD, Patty
Shandloff,
RN, and
Eduardo
Bancalari,
MD
From the Department of Pediatrics, Division of Neonatology, University of Miami School of Medicine, Miami
ABSTRACT. Anemia may increase the risk of tissue hy-poxia in preterm infants. This could lead to respiratory center depression and an increased risk for apnea. Heart
rate and breathing pattern were recorded in 30 preterm infants (gestational age 30.0 ± 2.3 weeks, postnatal age 46.6 ± 20.8 days, and weight 1,438 ± 266 g) before and
after a transfusion of 10 mL/kg of packed RBCs. All infants were stable clinically, breathing room air, and free of prolonged apneic episodes. After transfusion, he-matocrit levels increased from 27.0% ± 2.5% to 35.8% ±
4.7%. Heart rate decreased from 157.2 ± 13.6 beats per
minute to 148.4 ± 13.9 beats per minute. There was no
change in respiratory rate or BP. The duration of periodic breathing decreased significantly, as did the duration of
the longest periodic breathing episode (P < .01). The number of respiratory pauses lasting 5 to 10 seconds and the number of pauses lasting 11 to 20 seconds also
de-creased significantly (P < .05). The total duration of respiratory pauses, excluding pauses during periodic breathing, were significantly lower after transfusion (P < .05), as was the number of episodes of bradycardia. These results indicate that preterm infants have a more irregular breathing pattern while anemic than after
cor-rection of the anemia. The irregular breathing pattern is
probably caused by mild hypoxic respiratory center
depression. Pediatrics 1987;80:79-84; anemia, apnea, breathing pattern, hematocrit, preterm infant.
Physiologic anemia develops earlier and is
fre-quently more pronounced in the preterm infant
than in the infant born at term.’3 This reduction
in hemoglobin concentration is caused by several
mechanisms48 and occurs when the breathing
pat-tern of the infants is still characterized by frequent
Received for publication Jan 27, 1986; accepted Oct 7, 1986.
Reprint requests to (E.B.) Department of Pediatrics (R-131),
University ofMiami School ofMedicine, P0 Box 016960, Miami, FL 33101.
PEDIATRICS (ISSN 0031 4005). Copyright © 1987 by the
American Academy of Pediatrics.
periodic breathing and apneic episodes. Most
in-fants tolerate the reduction in hemoglobin
concen-tration well, but in some cases, tachycardia,
tach-ypnea, and lethargy associated with the decrease in
oxygen-carrying capacity can be observed.4 When
available oxygen is insufficient to meet the
meta-bolic demands, CNS hypoxia may occur, leading to
respiratory center depression and an increase in
periodic breathing and apnea.9” The possibility
that anemia may predispose to apnea is supported
by the clinical experience of healthy, growing pre-term infants who have tachycardia, tire during
feed-ing, show lethargy, and, finally, have prolonged
apneic episodes associated with a progressive
de-crease in hematocrit level. The development of
prolonged apnea is associated with an increase in
episodes of periodic breathing and an increasing number of short apneic episodes.’#{176}’3 It is difficult
to document the development of prolonged apnea
and its correlation with a gradually decreasing
he-matocrit level because of the large day to day
van-ability in breathing pattern’4 and the maturational
changes of the CNS and respiratory center, which
tend to counteract this development. Also, the
many stimuli and support measures that are
initi-ated as apnea develops will alter the breathing
pattern.
This study takes advantage of the rapid change
in hematocnit level occurring in preterm infants
who receive transfusions to correct low hematocnit
levels, to study breathing patterns free of long-term
maturational and environmental changes. It is
hy-pothesized that an increase in hematocnit level in
anemic preterm infants will lead to a stabilization
in breathing pattern, ie, a reduction in incidence of
periodic breathing and a decrease in the number of
apneic episodes. This stabilization would indicate
INFANTS AND METHODS
Infants were selected for this study from the
preterm patients of the convalescent nursery at
Jackson Memorial Hospital. An infant was entered
into the study after the attending physician had
ordered a transfusion for him or her. The decision
to transfuse was based on the routine neonatal care
guidelines and was not influenced by the study. The
patient received 10 mL/kg of packed RBCs for two
to three hours when the hematocrit level had
de-creased to 25% to 28%. Infants with higher
hema-tocrit levels were given transfusions if they showed
lethargy, poor feeding, tachycardia, or an increasing
number of apneic episodes.
The anticoagulant used by the blood bank was
citrate, phosphate, dextrose. The RBCs transfused, which had an age of five days or less, were washed three times in saline and packed. If an antibody screening test or antiglobulin test was negative,
type 0, Rh-negative uncrossmatched cells were
used. A crossmatch was done only when antibodies were present.
Heart rate and breathing pattern before and after
transfusion were recorded for 38 infants who were
entered into the study. Eight infants were excluded
from the analysis because they were receiving
ami-nophylline (n = 6), supplemental oxygen (n = 1),
or continuous positive airway pressure (n = 1). This
resulted in a clinically homogenous study group of 30 preterm infants who were breathing room air, were free of acute cardiorespiratory disease, were receiving oral feedings, and were growing normally.
Clinical problems that the infants had during their hospital course included hyaline membrane
disease (n = 16), apnea of prematurity (n = 14),
respiratory depression at birth (n = 9), patent duc-tus arteriosus (n = 7), sepsis (n = 6), subependymal/
intraventricular hemorrhage (n = 4), ventricular
dilation (n = 3), necrotizing enterocolitis (n = 2),
and chronic lung disease (n = 1). All these
condi-tions had resolved at the time of the study. The physical characteristics of the infants are given in
Table 1.
TABLE 1. Characteristics of 30 Preterm Infants Who
Received Transfusions
Clinical Characteristics Mean ± SD Range
Gestational age (wk) 30.0 ± 2.3 26-35
Postnatal age when 46.6 ± 20.8 18-105 studied (d)
Birth wt (g) 1,040 ± 235 700-1,560
Wt when studied (g) 1,438 ± 266 900-2,000
Apgar score
1 mm 4.2 ± 2.6 1-9
5 mm 7.2 ± 1.4 1-9
Sex (male/female) 12/18
Before and after transfusion, the hematocrit level
was determined from blood samples obtained by heel stick. Arterial BP was measured using an
au-tomated oscillometer (Dinamap Neonatal Vital
Signs Monitor, Critikon Inc, Tampa, FL). Heart rate and breathing pattern were recorded
immedi-ately before and after transfusion with a
Corome-tnics Infant Monitor (Corometrics Medical Systems
Inc, Wallingford, CT).
Only periods of quiet breathing were analyzed.
Excluded from the analysis were periods of activity
evident from the tracing by an elevated fluctuating
heart rate and by frequent changes in the baseline
of the breathing pattern, with the ventilatory
ex-cursions exceeding the range of the tracing. The
recordings were analyzed for the following events:
duration of periodic breathing, longest episodes of
periodic breathing, number of apneic episodes of 5
to 10 seconds in duration, number of apneic
epi-sodes of 1 1 to 20 seconds in duration, number of apneic episodes of >20 seconds in duration, total duration of apneic pauses, total number of brady-cardic episodes, and number of bradycardic epi-sodes without apnea.
Periodic breathing was defined as three or more
episodes of apnea lasting >3 seconds, interrupted
by breathing episodes of <20 seconds. The number
of apneic episodes and duration of apneic episodes
did not include apnea observed during periodic
breathing.
Bradycardia was defined as a decrease in heart
rate 25% below baseline that persisted longer than
5 seconds. Baseline heart rate was heart rate
ob-served during quiet regular breathing. All
respira-tory pattern measurements, except for the longest
episode of periodic breathing, were related to 100
minutes of recording time.
The average value for heart rate was calculated
by taking measurements every four minutes throughout the total length of the recording and averaging them. The average respiratory rate was
obtained from determinations made every one to
two hours during the 12 hours before and after transfusion.
The interpretation of the recordings was done by
two investigators. They were not blinded to which
recording was done before and which after
trans-fusion.
The Committee for the Protection of Human
Subjects at the University of Miami did not
con-sider informed consent necessary for the study.
Both the transfusion of RBCs given for specific
indications and the monitoring of heart rate and respirations were part of the routine care of preterm infants.
rate, and hematocrit measurement was done by calculating mean ± SD and by using the paired t
test to determine significance of differences
ob-served in the measurements before and after
trans-fusion. Because the respiratory measurements did not have a normal distribution, nonparametric analysis was used.’5
RESULTS
The average duration of recording was 5.7 ± 1.7
hours before and 5.3 ± 1.8 hours after blood
trans-fusion. The duration of recording ranged from 3.1 to 8.8 hours before and 2.7 to 8.2 hours after
trans-fusion. Hematocrit levels ranged from 24% to 31% before and from 31% to 46% after transfusion. The mean increase of 8.8% in hematocrit level after transfusion was significant, but there was no
sig-nificant change in arterial BP (Table 2).
Heart rate decreased significantly by a mean of
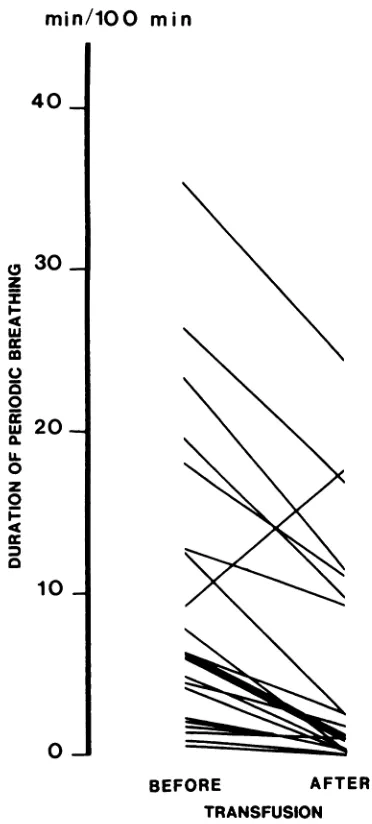
8.8 beats per minute and respiratory rate remained unchanged (Table 2). The measurements of breath-ing and heart rate pattern changed significantly after transfusion (Table 3). The most striking changes are shown in Figs 1, 2, and 3. Duration of periodic breathing decreased in 22 of the 30 infants and increased in only one. No periodic breathing was present both before and after transfusion in
seven infants (Fig 1). The decrease in median value
of periodic breathing was significant
(P
< .01). Thelongest episode of periodic breathing also decreased significantly after transfusion (Table 3).
The number of short apneic pauses lasting from
5 to 10 seconds decreased in 23 of 30 infants and
increased in seven (Fig 2). The median value of
short apneic pauses decreased significantly after transfusion (P < .05). The number of apneic pauses lasting 11 to 20 seconds, as well as the total duration
of respiratory pauses, also decreased after
transfu-sion. There were no respiratory pauses of more than 20 seconds’ duration before or after transfusion (Table 3).
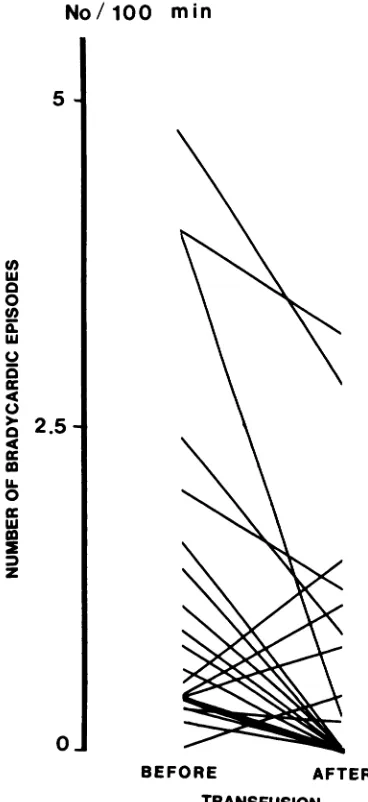
The total number of bradycardic episodes
de-creased in 15 of the 30 infants and increased in four
after transfusion (Fig 3). There was no bradycardia
either before or after transfusion in 11 infants. The
decrease in median value of bradycardic episodes
was significant
(P
< .05). There was also asignifi-cant decrease in bradycardic episodes not
associ-ated with apnea (Table 3).
None of the characteristics we measured (birth weight, gestational age, postnatal age, postnatal weight gain, hematocrit level, BP, or heart rate)
before and after transfusion could be identified as
more common among infants whose breathing
pat-tern did not improve after transfusion, compared
with those whose breathing pattern improved. There was no correlation between the severity of
anemia and the incidence of periodic breathing or
the number of apneic episodes. Also, the degree of
improvement after transfusion was not correlated
with initial hematocrit level. This finding is not
surprising because of the narrow range of
hemato-crit levels studied and the large interpatient
varia-tion in breathing pattern.
TABLE 2. Vital Signs and Hematocrit Levels Before and After Transfusion5
Before After P
Transfusion Transfusion Value
BP (mm Hg)
Systolic 73.6 ± 15.7 70.5 ± 12.6 NS
Diastolic 39.9 ± 10.8 40.1 ± 10.3 NS
Mean 52.2 ± 11.2 51.0 ± 10.5 NS
Heart rate (beats/mm) 157.2 ± 13.6 148.4 ± 13.9 .001
Respiratory rate (breaths/mm) 48.6 ± 12.2 49.8 ± 8.4 NS
Hematocrit (%) 27.0 ± 2.5 35.8 ± 4.7 .001
SResults are means ± SD.
TABLE 3. Respiratory Pattern Before and After Transfusion5
Respiratory Pattern Before After P
Transfusion Transfusion Value
Duration of periodic breathing (min/100 mm) 6.0 (0-35.1) 1.1 (0-24.3) .01
Longest episode of periodic breathing (mm) 3.1 (0-27.5) 1.4 (0-17.0) .01
No. of respiratory pauses 5-10 s/100 mm 9.0 (1.7-26.0) 5.0 (0-20.7) .05
No. of respiratory pauses 11-20 s/100 mm 0 (0-1.1) 0 (0-0.4) .05
No. of respiratory pauses >20 s/100 mm 0 0
Duration of respiratory pauses (min/100 mm) 0.9 (0.2-3.2) 0.4 (0-2.1) .05 No. of bradycardic episodes/100 mm 0.4 (0-4.8) 0.0 (0-3.2) .05 No. of bradycardic episodes without apnea/100 mm 0.3 (0-3.6) 0.0 (0-3.2) .01
min/100 mm
40_
3O_ z I I-4 Ui
U
0
Ui a. U. 0 z 0
4
10.
0-BEFORE AFTER
TRANSFUSION
Fig 1. Changes in duration of periodic breathing before and after transfusion of individual patients. Heavy line indicates median change (P < .01).
DISCUSSION
Anemia in the preterm infant may predispose to
tissue hypoxia because of reduced O2-carrying
ca-pacity of the blood. Tissue oxygenation may be further compromised because of the left shift of the 02 dissociation curve due to the presence of fetal hemoglobin. This makes the unloading of oxygen more difficult at the tissue level.4’6”7 The preterm
infant’s ability to compensate for hypoxia with an
increased cardiac output is limited,’8 and these in-fants frequently have impaired pulmonary function because of immature lungs or chronic lung disease.
In the present study, transfusion led to a signif-icant decrease in heart rate, suggesting that the increased heart rate before transfusion was a
re-sponse to the anemia, producing a compensatory increase in cardiac output.4
Hypoxia is an important cause of respiratory
depression in the newborn infant and is associated
with an increased incidence of periodic breathing
and apneic episodes.9”#{176}” Relieving hypoxia may stabilize the respiratory pattern and lead to a
re-duction in periodic breathing and apnea.’9 Anemia
severe enough to produce tissue hypoxia has been
associated with reduced central respiratory drive in animals2#{176} and with apnea in preterm infants.2”22
The infants studied here were a homogeneous
group of preterm growing infants, and all were in stable clinical condition without signs of distress or
evidence of prolonged apnea. However, they showed
an increased incidence of periodic breathing, short
apneic episodes, and bradycardia that improved
after transfusion. The findings suggest that before
transfusion these infants had some respiratory cen-ter depression, which was probably caused by mild CNS hypoxia.
More severe anemia than in the infants of this
No
/1
00
mm
30
C-) ‘U Cl) 0
I-I’)
C/) ‘U
0
-0 Cl) 0. ‘U
C-)
‘U
z
410-0
BEFORE
AFTER
TRANSFUSION
Fig 2. Changes in number of short apneic episodes
crystalloid infusion is not clear. However, in the present study, there was no evidence of hypotension
before the transfusion and no BP increase
after-ward, suggesting that the infants were not
hypo-volemic.
Prolonged apneic episodes that may be triggered
by anemia can usually be detected by cardiorespi-ratory monitoring and terminated before severe tissue hypoxia occurs. The well-documented risks
of transfusion (transmission of viral diseases,
in-compatibility, and immunologic consequences)
need to be kept in mind and weighed against the
possible benefits of keeping a higher hematocrit
level in healthy preterm infants. Further work
needs to be done before a change in the indications
for blood transfusion in small preterm infants can
be recommended. No/100 mm 5 Cl) UI 0 0 Cl) a. UI C.) 0 4 C.) 0 4 U. 0 UI z 0 BEFORE AFTER TRANSFUSION
Fig 3. Changes in number of bradycardic episodes be-fore and after transfusion of individual patients. Heavy line indicates median change (P < .01).
study, an increased metabolic rate, reduced ability to increase cardiac output, or a mild hemoglobin desaturation may lead to more severe respiratory
center depression and prolonged apnea. This was
not seen in the infants studied here because we tried to keep their hematocrit levels above 25% and because infants with active medical problems were
excluded from the study.
It is possible that the painful stimulus of
insert-ing an IV catheter had some effect on respiratory
drive. It is, however, unlikely that this effect lasted
for more than two to three hours, by which time transfusion was completed and the pneumogram recording was started. The fact that heart rates decreased after transfusion also argues against the
possibility that breathing pattern improved because
of physical stimulation. Whether or not the same changes in breathing pattern might be obtained by
simple blood volume expansion using colloid or
ACKNOWLEDGMENT
This work was supported, in part, by the State of Florida, Department of Health and Rehabilitative
Serv-ices, Children’s Medical Services, and The University of Miami: Project Newborn.
REFERENCES
1. O’Brian RT, Pearson HA: Physiologic anemia of the new-born. J Pediatr 1971;79:132-138
2. Schulman J: Anemia of prematurity. J Pediatr 1959;54:663-672
3. Stockman JA III: Anemia of prematurity. Clin Perinatol
1977;4:239-257
4. Wardrop CAJ, Holland BM, Veale KEA, et al:
Nonphysio-logic anemia of prematurity. Arch Dis Child 1978;53:855-860
5. Bratteby LE, Garby L, Groth T: Studies on erytho-kinetics in infancy: XIII. The mean life span and the life span frequency function of red blood cells formed during foetal
life. Acta Paediatr Scand 1968;57:311-320
6. Bratteby LE: Studies on erythrokinetics in infancy: XI. The change in circulating red cell volume during the first five months of life. Acta Paediatr Scand 1968;57:215-224
7. Brown MS, Garcia JF, Phibbs RH, et a!: Decreased response of plasma immunoreactive erythropoietin to “available ox-ygen” in anemia of prematurity. J Pediatr 1984;105:793-798
8. Stockman JA III, Graeber JE, Clark DA, et a!: Anemia of
prematurity: Determinants of the erythopoietin response. J Pediatr 1984;105:786-792
9. Cross KW, Opp#{233}TE: The effect of inhalation of high and low concentrations of oxygen in the respiration of the pre-mature infant. J Physiol 1952;117:38-55
10. Rigatto H, Brady JP: Periodic breathing and apnea in
pre-term infants: II. Hypoxia as primary event. Pediatrics
1972;50:219-228
11. Rigatto H, Brady JP, de la Tone Verduzco R:
Chemorecep-tor reflexes in preterm infants: I. The effect of gestational
and postnatal age on the ventilatory response to inhalation
of 100% and 15% oxygen. Pediatrics 1975;55:604-613 12. Daily WJR, Klaus M, Meyer HBP: Apnea in premature
infants: Monitoring, incidence, heart rate changes, and an
effect of environmental temperature. Pediatrics 1969;43: 510-581
13. Steinschneider A: Prolonged sleep apnea and respiratory
instability: A discriminative study. Pediatrics
1977;59:962-970
14. Hunt CE, Brouillette RT, Lin K, et a!: Day to day
pneu-mogram variability. Pediatr Res 1985;19:174-177
Sta-tistics. Danville, IL, Interstate Printers and Publishers Inc,
1959
16. Woodson RD: Physiologic significance of oxygen
dissocia-tion curve shifts. Crit Care Med 1979;7:368-373
17. Oski FA: Clinical implications of the oxyhemoglobin disso-ciation curve in the neonatal period. Crit Care Med 1979; 7:412-418
18. Romero TE, Friedman WF: Limited left ventricular
re-sponse to volume overload in the neonatal period: A corn-parative study with the adult animal. Pediatr Res 1979;
13:910-915
19. Rigatto H, Brady JP: Periodic breathing and apnea in
pre-term infants: I. Evidence for hypoventilation possibly due to central respiratory depression. Pediatrics 1972;50:202-218
20. Fagenholz SA, Lee JC, Downing SE: Association of anemia
with reduced central respiratory drive in the piglet. Yak J
Biol Med 1979;52:263-270
21. Wertharnrner J, Kelly DH, Shannon DC: Efficacy of blood
transfusion in reducing apnea in anemic premature infants, abstracted. Pediatr Res 1983;17:341A
22. DeMaio JG, Harris MC, Spitzer AR: The response of apnea of prematurity to transfusion therapy, abstracted. Pediatr Res 1986;20:389A
WHAT AMERICAN
CHILDREN
READ IN 1834 ABOUT
PAST INGRATITUDES
TOWARD
THEIR
PARENTS
The Reverend John S. C. Abbott of Worcester, Massachusetts, a respected
early nineteenth century author of books on family morals and duties, warned
children about past ingratitudes toward their parents as follows:
And when your mother dies, do you not think that you will feel remorse for every
unkind word you have uttered, and for every act of ingratitude? Your beloved parents must soon die. You will probably be led into their darkened chamber, to see them pale
and helpless on their dying bed. Oh, how will you feel in that solemn hour! All your past life will come to your mind, and you will think that you would give worlds, if you could
blot out the remembrance of past ingratitude. You will think that, if your mother or father should only get well, you would never do any thing to grieve them again. But the hour for them to die must come. You may weep as though your heart would break, but it will not recall the past, and it will not delay their death. They must die; and you will probably gaze upon their cold and lifeless countenances in the coffin. You will follow
them to the grave, and see them buried forever from your sight. Oh, how unhappy you will feel, if you then have to reflect upon your misconduct! The tears you will shed over their graves will be the more bitter, because you will feel that perhaps, your own
misconduct hastened their death.
REFERENCE
Noted by T.E.C., Jr, MD