Original Article
Association between
ACE
polymorphisms and
osteoarthritis susceptibility
Zhong Qing, Jiumin Ye
Honghui Hospital Affiliated to Medical College of Xi’an Jiao Tong University, Friendship Road, Xi’an, China
Received February 17, 2015; Accepted April 13, 2015; Epub June 1, 2015; Published June 15, 2015
Abstract: Objective: The present study was designed to investigate the association ofangiotensin-converting en-zyme (ACE) rs4343 and rs4362 polymorphisms with the susceptibility to osteoarthritis (OA). Methods: 109 knee OA patients and 114 healthy people were enrolled in the study. Polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) was used to perform the genotyping for two groups and the linkage disequilibrium and haplotype were analyzed using Haploview software. The differences of genotype and allele frequencies were analyzed by χ2 test and Fisher’s exact test. The relationship between ACE polymorphisms and OA susceptibility was represented by odds ratios (ORs) with 95% confidence intervals (95% CIs). Results: The genotypes distributions of
ACE rs4343 and rs4362 polymorphisms in control groups were accordance with HWE. ACE rs4343 polymorphism was associated with the significantly increased risk of OA (AG vs. AA: OR=2.41, P=0.003; GG vs. AA: OR=5.35,
P=0.015; G vs. A: OR=2.27, P<0.001). Similarly, rs4362 polymorphisms was also a risk factor for OA (CT vs. CC: OR=2.60, P=0.005; TT vs. CC: OR=3.15, P=0.003; T vs. C: OR=1.88, P=0.001). The result of haplotype analysis showed complete linkage disequilibrium in rs 4343 and rs 4362 polymorphisms. The G-T haplotype significantly increased OA susceptibility, but A-C is a protective factor for the occurrence of OA. Conclusion: Significant correlation exists between ACE rs4343and rs4362 polymorphisms and OA. In haplotype analysis, A-C haplotype may provide protection against OA, and G-T haplotype may be a risk factor for the development of OA.
Keywords:ACE, osteoarthritis, polymorphism, risk
Introduction
Osteoarthritis (OA) is a common disease of joint surgery that can lead to the loss of joint func-tion [1]. The pathological change of OA involves all structures of joint, especially cartilage [2]. OA can cause the arthralgia, malformation and dysfunction of joint, the sclerosis of subchon-dral bone, joint space narrowing, syndesmoph-yte formation, synovitis and the contracture or
flab of ligament [3, 4]. The worst result makes
the patients disabled and affects the quality of life seriously [5, 6]. Understanding the
patho-logical mechanism of OA is the only way to find
effective methods for preventing and reducing the occurrence of OA.
Epidemiological studies point out that OA is a complex disease involving combined actions of genetic and environment factors as well as mul-tiple other factors [7, 8]. Age, hormone, and trauma are all a relationship with the
occur-rence and development of OA [9]. Additionally, OA has been shown in studies to have a strong genetic predisposition and the pathogenesis may be associated with some genetic polymor-phisms [10, 11].
has proved that ACE has I/D (rs4340) polymor-phism associated with theexpressionlevel and activity of ACE protein in human body [15]. McKenzie et al. pointed out that ACE rs4343 polymorphism on extron 17 is highly related to the expression level of ACE [16].
In recent years, the relationship between ACE
polymorphisms and diseases has become a
hot spot in research field. However, most of
studies focus on the pathogenesis of hyperten-sion, diabetes and cardiovascular diseases
[17-20]. Poornima et al. have certified that ACE
polymorphisms are closely related to type 2 diabetes and gestational diabetes mellitus [21]. Meanwhile, Narne et al. also suggested that the genetic variant of ACE played impor-tant roles in coronary artery disease (CAD) and myocardial infarction (MI) [22]. The previous studies, however, choose one single polymor-phism (mainly rs4340) as the target, being dis-advantageous to comprehensive study. Our study explored two polymorphisms of ACE
rs4343 and rs4362 to investigate the associa-tion between ACE polymorphisms and OA sus-ceptibility, which provides theoretical basis for further exploration of OA pathogenesis and the early prevention.
Materials and methods
Subjects
All subjects including 109 knee OA patients and 114 healthy people were recruited from Xi’an Honghui Hospital. The OA patients were
confirmed by clinical examination and radio -graphic inspection. Patients were excluded if they suffered from other kinds of arthritis such as traumatic arthritis (TA), infective arthritis and skeletal dysplasia, or had tumor history. According to the Kellgren/Lawrence standard, patients with a score of 2 or more would be enrolled in the case group. The healthy controls without the history of tumors, immunopathy and osteoarthritis disease were collected from the physical examination center of the same hospital. They were frequency-matched with cases by age, gender and body mass index (BMI). To ensure the accuracy of the study, all subjects belonged to Chinese Han population for a long time and were not related by blood. Our research obtained the approval from the Ethics Committee and written informed con-sent from each participant.
Collection of clinical data and sample from study subjects
The clinical information of all subjects in two groups were collected, including age, gender, onset age, weight, stature and body mass index
(BMI= Weight (kg)/Height²), especially the his -tory of relevant diseases, such as hyperten-sion, diabetes and thyroid dysfunction.
5 ml fasting peripheral venous blood from every subject was collected in EDTA anticoagulant
tubes. Genome DNA was extracted with DNA
extraction kit according to manufacturer instructions. DNA samples were conducted the test of quality and concentration with ultravio-let spectrophotometer, and preserved at -20°C refrigerator.
Genotyping of ACE polymorphisms
PCR primers of ACE rs4343 and rs4362 poly-morphisms were designed using Primer pre-mier 5.0 software and synthesized by Shanghai Sangon Biological Engineering Technology Service Co., LTD. The PCR reactions system
was a volume of 25 μL solution, including 1 μL DNA template substrates, 12.5 μL Master Mix (2×), each 0.5 μL forward and reverse primers
and was added with aseptic dd H2O to 25 μL.
PCR conditions were as follows: initial denatur-ation at 95°C for 10 min, followed by 30 cycles of denaturation at 95°C for 15 s, annealing at 60°C for 50 s, extending at 72°C for 30 s and
final extension at 40°C for 5 min. PCR products
were digested by restriction enzyme Hal III and
MboI,2% agarose gel electrophoresis was con-ducted to separate these restriction frag- ments.
Statistical analysis
Experimental data were shown as _x±SD. The representativeness of genotypes in the control group was estimated by Hardy-Weinberg equi-librium (HWE). The differences of genotype and
allele distributions were analyzed by χ2 test and
Fisher’s exact test, and represented by odds
ratios (ORs) and 95% confidence intervals (CIs).
Statistical analysis was conducted by SPSS 18.0 software. Haploview software was used to calculate the linkage disequilibrium and haplo-type composition among SNPs with statistical
Results
Comparison of clinical data of the subjects
The clinical data of all subjects are shown in Table 1. The average BMI of 109 cases (47 males, 62 females) was 26.52±3.74 kg/m2.
Their median age was 54.25±9.75 while the average onset age was 42.54±7.28. The BMI of 114 controls (51 males, 63 females) was 24.27±2.78 kg/m2. Their average age was
52.13±8.59. No statistically significant differ -ences existed in age, sex and BMI between two
CC: OR=2.60, 95% CI=1.37-4.93; TT vs. CC: OR=3.15, 95% CI=1.49-6.68; T vs. C: OR=1.88, 95% CI=1.29-2.74). Compared with T allele in rs4362, G in rs4343 was more likely to be the
susceptible allele to OA.
Linkage disequilibrium and haplotype analysis
Linkage disequilibrium analysis showed that complete linkage disequilibrium existed be- tween ACE rs4343 and rs4362 (D’=1.0, r2
=0.832). Further analysis of the frequencies
[image:3.612.91.357.85.215.2]difference of established haplotypes between Table 1. Comparison of clinical information of the subjects
Clinic information (n=109)Case (n=114)Control P
Age 54.25±9.75 52.13±8.59 0.765
Sex (male: female) 47:62 51:63 0.893
BMI (kg/m2) 26.52±3.74 24.27±2.78 0.698
Onset age 42.54±7.28 —
Disease history
[image:3.612.92.357.263.379.2]Hypertension [n (%)] 41 (37.61) 11 (9.65) <0.001 Diabetes [n (% )] 32 (29.36) 9 (7.89) <0.001 Thyroid dysfunction [n (%)] 35 (32.11) 18 (15.79) 0.005
Table 2. The genotype and allele distribution comparison of
ACE gene rs4343 polymorphism of the subjects
rs4343 (n=109, %)Case (n=114, %)Control χ2 P OR (95% CI) Genotype
AA 48 (44.0) 77 (67.6) - - 1.00
AG 51 (46.8) 34 (29.8) 9.47 0.003 2.41 (1.37-4.23) GG 10 (9.2) 3 (2.6) 7.17 0.015 5.35 (1.40-20.4) Allele
A 147 (67.4) 188 (82.5) - - 1.00
G 71 (32.6) 40 (17.5) 13.5 <0.001 2.27 (1.46-3.54)
groups (P>0.05). However, signifi -cant differences were exposed in such aspects as hypertension, dia-betes and thyroid dysfunction (P<0.05) through the investigation of participants’ disease history. Data indicated that the onset of OA had a relationship with hyperten-sion, diabetes and immune organ diseases.
HWE test
The HWE test showed the geno- type distributions of ACE rs4343 and rs4362 polymorphisms we- re consistent with the law of ge- netic equilibrium in control gro-
up (rs4343: χ2=0.109, P=0.742;
rs4362: χ2=2.521, P=0.112),
whi-ch indicated that our subjects were randomly selected and had de- mographic representativeness.
Correlation analysis of ACE poly
-morphisms and OA risk
The genotype and allele distribu-tions of ACE rs4343 and rs4362 polymorphisms in two groups are shown in Tables 2 and 3. The geno-type and allele distributions of rs4343 and rs4362 in two groups
were significantly different (P<
0.05) indicating that G allele at
rs4343 could increase the risk of
OA (AG vs. AA: OR=2.41, 95% CI=1.37-4.23; GG vs. AA: OR=5.35, 95% CI=1.40-20.4; G vs. A: OR=2.27, 95% CI=1.46-3.54) and
[image:3.612.93.356.426.544.2]T allele at rs4362 might be the susceptible factor for OA (CT vs. Table 3. The genotype and allele distribution comparison of
ACE gene rs4362 polymorphism of the subjects
rs4362 (n=109, %)Case (n=114, %)Control χ2 P OR (95% CI) Genotype
CC 22 (20.2) 47 (41.2) - - 1.000
CT 56 (51.4) 46 (40.4) 8.79 0.005 2.60 (1.37-4.93) TT 31 (28.4) 21 (18.4) 9.26 0.003 3.15 (1.49-6.68) Allele
C 100 (45.9) 140 (61.4) - - 1.00
two groups manifested that there were 3 haplo-types formed by rs4343 and rs4362: A-C, A-T
and G-T. The haplotype frequencies difference of A-C and G-T between two groups had statisti
-cal significance (P<0.05). The result from the study indicated that ACE haplotype A-C might
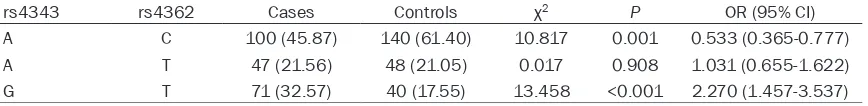
offer protection against OA (OR=0.533, 95% CI=0.365-0.777), and that G-T haplotype might increase the risk of OA (OR=2.270, 95% CI=1.457-3.537) (Table 4).
Discussion
Osteoarthritis (OA) is a degenerative joint dis-ease caused by a variety of factors, and there are tens of millions of OA patients waiting for cure [23]. However, the pathogenesis of OA has not entirely clear yet. Studies in recent years
have proved that inflammation plays an impor -tant role for the occurrence of OA and ACE is closely associated with rheumatic and autoim-mune diseases [24-26]. Therefore, the present research explored the relationship of ACE
rs4343 and rs4362 polymorphisms with the susceptibility to OA to provide certain theoreti-cal basis for prevention and treatment.
ACE (about 21 kb) is located on chromosome 17 q23, containing 26 exons and 25 introns [27]. Although ACE has many polymorphisms, most studies focus on the insert and/or dele-tion (I/D) fragment polymorphisms of the 16th
intron widely. Two new SNPs of ACE were explored in the present study. Rs4343 poly-morphism in ACE exon 17 has been certified by
previous studies to have some correlation with essential hypertension, heart disease, ne- phropathy and Alzheimer disease [28-30], but there was no report on its association with osteoarthritis. Additionally, rs4362 polymor-phism of ACE exon 24 had not been reported its relationship with osteoarthritis either. Our study showed that the differences between knee osteoarthritis patients and healthy con-trols in disease history including hypertension, diabetes and immune organ diseases had
sta-tistical significance (P<0.05), indicating that
the development of OA was related to hyperten-sion, diabetes and immune organ function. The genotype and allele distributions of rs4343 and rs4362 polymorphisms demonstrated
sig-nificant differences between cases and con -trols (P<0.05). The rs4343 G allele could
increase OA risk, similarly, the rs4362 T allele might be the susceptible factor for OA. The fur-ther haplotype analysis of rs4343 and rs4362 polymorphisms suggested that A-C haplotype might have a protective effect against the
inci-dence of OA, but G-T haplotype might increase
the risk of OA.
The rs4343 and rs4362 polymorphisms are located in exon region of ACE, and both of them are synonymous mutations. The replacement of bases does not change amino acid composi-tion in ACE. However, McKenzie et al. demon-strated that rs4343 polymorphism could affect the expression level of ACE in serum or tissues
and G allele could lead to a significant change
in ACE expression level. Therefore, it was inferred that ACE rs4343 and rs4362 polymor-phisms might affect the susceptibility to OA by
influencing the expression level of ACE in serum or tissues. Besides, our research combined two polymorphisms of rs4343 and rs4362 in ACE
gene to evaluate their relationship with the progress and occurrence of OA susceptibility, so it was more convincing than one single poly-morphism. The polymorphisms of ACE rs4343 and rs4362 may participate jointly in the patho-physiology of OA, but the exact mechanism needs to be further explored.
Disclosure of conflict of interest None.
Address correspondence to: Dr. Zhong Qing, Honghui Hospital Affiliated to Medical College of Xi’an Jiao Tong University, Friendship Road, Xi’an, China. E-mail: qingzhng@tom.com
References
[1] Forestier R, Francon A, Briole V, Genty C, Chevalier X and Richette P. Prevalence of gen-Table 4. Haplotype frequency comparison of ACE rs4343 and rs4362 polymorphisms of the subjects
rs4343 rs4362 Cases Controls χ2 P OR (95% CI)
A C 100 (45.87) 140 (61.40) 10.817 0.001 0.533 (0.365-0.777)
A T 47 (21.56) 48 (21.05) 0.017 0.908 1.031 (0.655-1.622)
[image:4.612.91.525.86.139.2]eralized osteoarthritis in a population with knee osteoarthritis. Joint Bone Spine 2011; 78: 275-278.
[2] Horton WE Jr, Bennion P and Yang L. Cellular, molecular, and matrix changes in cartilage during aging and osteoarthritis. J Musculos- kelet Neuronal Interact 2006; 6: 379-381. [3] Straw R, Kulkarni S, Attfield S and Wilton TJ.
Posterior cruciate ligament at total knee re-placement. Essential, beneficial or a hin-drance? J Bone Joint Surg Br 2003; 85: 671-674.
[4] Ranawat CS, Insall J and Shine J. Duo-condylar knee replacement. Curr Pract Orthop Surg 1975; 6: 28-35.
[5] Khatod M, Inacio M, Paxton EW, Bini SA, Namba RS, Burchette RJ and Fithian DC. Knee replacement: epidemiology, outcomes, and trends in Southern California: 17,080 replace-ments from 1995 through 2004. Acta Orthop 2008; 79: 812-819.
[6] Simmons S, Lephart S, Rubash H, Borsa P and Barrack RL. Proprioception following total knee arthroplasty with and without the posterior cru-ciate ligament. J Arthroplasty 1996; 11: 763-768.
[7] Peach CA, Carr AJ and Loughlin J. Recent ad-vances in the genetic investigation of osteoar-thritis. Trends Mol Med 2005; 11: 186-191. [8] Arden N and Nevitt MC. Osteoarthritis:
epide-miology. Best Pract Res Clin Rheumatol 2006; 20: 3-25.
[9] Bae JY, Park KS, Seon JK, Kwak DS, Jeon I and Song EK. Biomechanical analysis of the ef-fects of medial meniscectomy on degenerative osteoarthritis. Med Biol Eng Comput 2012; 50: 53-60.
[10] Anderson DD, Marsh JL and Brown TD. The pathomechanical etiology of post-traumatic osteoarthritis following intraarticular fractures. Iowa Orthop J 2011; 31: 1-20.
[11] Michou L. Genetics of digital osteoarthritis. Joint Bone Spine 2011; 78: 347-351.
[12] Brzozowski T. Role of renin-angiotensin system and metabolites of angiotensin in the mecha-nism of gastric mucosal protection. Curr Opin Pharmacol 2014; 19: 90-98.
[13] Johnston CI. Franz Volhard Lecture. Renin-angiotensin system: a dual tissue and hormon-al system for cardiovascular control. J Hypertens Suppl 1992; 10: S13-26.
[14] Freire MB, van Dijk DJ, Erman A, Boner G, Warram JH and Krolewski AS. DNA polymor-phisms in the ACE gene, serum ACE activity and the risk of nephropathy in insulin-depen-dent diabetes mellitus. Nephrol Dial Transplant 1998; 13: 2553-2558.
[15] Lu XM, Chen GJ, Yang Y and Qiu HB. Angiotensin-converting enzyme polymorphism
affects outcome of local Chinese with acute lung injury. Respir Med 2011; 105: 1485-1490.
[16] McKenzie CA, Sinsheimer JS, Adeyemo AA, Cox RD, Southam L, Hugill A, Bouzekri N, Lathrop M, Forrester TE, Cooper RS and Ward R. SNP haplotypes in the angiotensin I-converting en-zyme (ACE) gene: analysis of Nigerian family data using gamete competition models. Ann Hum Genet 2005; 69: 227-232.
[17] Ruiz J, Blanche H, Cohen N, Velho G, Cambien F, Cohen D, Passa P and Froguel P. Insertion/ deletion polymorphism of the angiotensin-con-verting enzyme gene is strongly associated with coronary heart disease in non-insulin-de-pendent diabetes mellitus. Proc Natl Acad Sci U S A 1994; 91: 3662-3665.
[18] Morise T, Takeuchi Y and Takeda R. Angiotensin-converting enzyme polymorphism and essen-tial hypertension. Lancet 1994; 343: 125. [19] Schunkert H, Hense HW, Holmer SR, Stender
M, Perz S, Keil U, Lorell BH and Riegger GA. Association between a deletion polymorphism of the angiotensin-converting-enzyme gene and left ventricular hypertrophy. N Engl J Med 1994; 330: 1634-1638.
[20] Samsonov M, Lopatin J, Tilz GP, Artner-Dworzak E, Nassonov E, Mareev V, Belenkov J, Wachter H and Fuchs D. The activated immune system and the renin-angiotensin-aldosterone system in congestive heart failure. J Intern Med 1998; 243: 93-98.
[21] Poornima S, Subramanyam K, Khan IA and Hasan Q. The insertion and deletion (I28005D) polymorphism of the angiotensin I converting enzyme gene is a risk factor for osteoarthritis in an Asian Indian population. J Renin Angiotensin Aldosterone Syst 2014; [Epub ahead of print].
[22] Narne P, Ponnaluri KC, Singh S, Siraj M and Ishaq M. Relationship between angiotensin-converting enzyme gene insertion/deletion polymorphism, angiographically defined coro-nary artery disease and myocardial infarction in patients with type 2 diabetes mellitus. J Renin Angiotensin Aldosterone Syst 2012; 13: 478-486.
[23] de Grandmont P. Osteoarthrosis/osteoarthritis in the temporomandibular joints. Int J Prosthodont 2009; 22: 530-532.
[24] Banks SE. Erosive osteoarthritis: a current re-view of a clinical challenge. Clin Rheumatol 2010; 29: 697-706.
[25] Loeser RF. Aging and osteoarthritis: the role of chondrocyte senescence and aging changes in the cartilage matrix. Osteoarthritis Cartilage 2009; 17: 971-979.
synovium: increased stromal [(125)I]351A binding in rheumatoid arthritis. Ann Rheum Dis 2000; 59: 125-131.
[27] Sayed-Tabatabaei FA, Oostra BA, Isaacs A, van Duijn CM and Witteman JC. ACE polymor-phisms. Circ Res 2006; 98: 1123-1133. [28] Pan M, Zhu JH, Liu ZH, Jiang WP, Cui ZC, Yu XH,
Li HM and Yang XJ. Angiotensin-converting en-zyme gene 2350 G/A polymorphism is associ-ated with left ventricular hypertrophy but not essential hypertension. Hypertens Res 2007; 30: 31-37.
[29] Wenquan N, Yue Q, Pingjin G and Dingliang Z. Review: association between angiotensin con-verting enzyme G2350A polymorphism and hypertension risk: a meta-analysis. J Renin Angiotensin Aldosterone Syst 2011; 12: 8-14.