BSOVE
13(
2):
221- 360 ( 1988)
ISSN 0146- 6429
Bulletin of the
SOCIETY FOR
VECTOR ECOLOGY
BULLETIN OF THE
SOCIETY FOR VECTOR ECOLOGY
Volume 13- Number 2 December 1988James P. Webb, Editor
Orange County Vector Control District
13001 Garden Grove BoulevardGarden Grove, CA 92643
EDITORIAL BOARD
Mir S. Mulla, Chairman,
University of California
Riverside, CA 92521
A.J.Adames J.B.Davies R. S.Lane F. J. Radovsky
University of Panama Liverpool Sch. ofTropMedicine UniversityofCalifornia NC State Museum Nat. Hist.
Panama Liverpool, England Berkeley,California Raleigh, North Carolina
A.All M. S. Dhillon L. A. Magnarelli R. E. Ryckman
University of Florida Northwest M.A.D. Conn. Agr. Exp. Station Loma Linda University Sanford, Florida Riverside, California New Haven, Connecticut Loma Linda, California
D.R. Barnard P.Eisen C.J.Mitchell H. I. Scudder
Lone Star Tick Research Lab. Institute of Trop. Medicine Centers for Disease Control California State University
Poteau, Oklahoma Antwerp, Belgium Fort Collins, Colorado Hayward, California
N. Becker R. E.Fontaine G. R.Mullen M. W. Service
University of Heidelberg CDC, Public Health Service Auburn University Liverpool Sch. of Trop. Mcd. Heidelberg, West Germany Atlanta, Georgia Auburn, Alabama Liverpool, England
H. Briegel M. L. GoIT S. Salim E. S. Tikasingh
University of Zurich University of Hawaii, Manoa VectorControlResearch Unit Caribbean Epidemiology Ctr. Zurich, Switzerland Honolulu, Hawaii Jakarta, Indonesia Port of Spain, Trinidad
G. G.Clark D.J.Gubler G. F.O' Meara G. B. White
CDC, PublicHealth Service CDC, Public Health Service University of Florida, WAS London Sch. Hyg. Trop. Mcd. San Juan, Puerto Rico San Juan, Puerto Rico Vero Beach, Florida London, England
W.J.Crans C. FHopla S.Palchick S. C. Williams
Rutgers University
University of Oklahoma Metro. Mosq. Cont. Dist. San Francisco State Univ.
New Brunswick, New Jersey Norman, Oklahoma St. Paul, Minnesota San Francisco, California
C.Dahl M.J.Klowden M. A. Parsons W. J. Wrenn
Uppsala University
University of Idaho Department of Health University of North Dakota
Uppsala,Sweden Moscow, Idaho Columbus, Ohio Grand Forks, North Dakota
L. A.Lacey D. B. Pence
Medical Services Consultants Texas Tech University
Arlington, Virginia Lubbock, Texas
Published by the Society for Vector Ecology to disseminate pertinent information from all facets of the field of Vector Ecology and Related Disciplines
Membership
Plus Bulletin $ 25. 00Student
Membership $
12. 50 Institutional Subscription $ 25.00Sustaining
Members $ 150. 00Communications relating to editorial matters and manuscripts should be addressed to the Editor. Communications relating to Balky proofs,
reprints, subscriptions, SOVE membership, change of address, and other matters
should be addressed tothe Business Omen. PUBLICATIONS AND BUSINESS OFFICE
Society for Vector Ecology P. O. Box 87
Santa Ana, California 92702 USA
PrintingConsultant Minoo B. Madon, Production Manager
John G. Shanafelt, Jr. Environmental Management Branch
Orange, California California State Department of Health Services
BULLETIN
OF THE
SOCIETY FOR VECTOR ECOLOGY
Volume 13 December, 1988 Number 2
CONTENTS
Submitted Papers
The Aquatic Insect Communities of Tree Holes in Northern California Oak Woodlands
D.L.Woodward, A. E. Colwell, andN.L. Anderson 221
Population Trends and Behavioral Attributes of Adult Mosquitoes Associated with Dairies in Southern
California E. T. Schreiber,M.S. Mulla,andJ. D.
Chaney
235Factors Affecting Oviposition and Egg Production in Laboratory- Reared Anopheles pharoensis
Theobald A.S. El- AkadandJ. G.Humphreys 243
The Effect of Habitat Management and Toxic Bait Placement on the Movement and Home Range
Activities of Telemetered Rattus rattus in Orange County, California
M. A. Recht, R.Geck,G.L.Challet,andJ. P. Webb 248
Proceedings
2nd European SOVE Branch Meeting, Heidelberg, WEST GERMANY
31 August-2 September 1987
Black Fly ( Diptera: Simuliidae) Control in West Germany with the Biological Larvicide Bacillus
thuringiensisvar.israelensis W. E. Deschle, H.-E. Hagen, J. Rutschke,M. Starner,andT. Meyer 280
19th Annual SOVE Conference, Asilomar Conference Center, Pacific Grove, California
18-20 November 1987Current Trends in Researchand
Training
inVector- Borne Diseaseon aGlobal Basis T. Godal 287Urban Vectorand Pest Control in
Developing
Countries R.J. Tonn 291ControlPotentials in
Feeding
MechanismsofMosquito Larvae C. Dahl 295Aedesaegypti:
Why
Can'tWe Control It? S. B. Halstead 304CantheWartoContainVectors be Lost? B. H.
Kay
312Marking Adult Mosquitoes Using Fluorescent Pigments in Dispersal Studies
C. L. Meek, J. C. Fryer,andM. L.Niebylski 319
Medical
Entomology
Manpower: AnAnalysisandaPlan H. I.Scudder 323Observations on Anopheline and Malaria Ecology in the Far Western Region of Nepal, 1986
S. L. Shrestha, S. Pradhan, J. P. B. Shrestha,
J. D. Shrestha, Y. Rajbhamdari, G. L. Shrestha, T. B. Swar, M. K.Nushin, and W. K. Reisen 332
Performance of Pyrethroids Against German Cockroaches Blattella germanica (L.) (Dictyoptera:
Blattellidae) M. K.RustandD. A.Reierson 343
Bluetongue in the United States: Status, Transmission, and Control Through Vector Suppression
F. R. Holbrook 350
VectorControlonU. S.
Army
Installations R. N.Johnson 354BULL. SOC. VECTOR ECOL., 13( 2): 221- 234
DECEMBER, 1988
THE AQUATIC INSECT COMMUNITIES OF TREE HOLES IN
NORTHERN CALIFORNIA OAK WOODLANDS
D. L. Woodward', A. E. Colwell', and N. L. Anderson'
ABSTRACT: A survey of the aquatic insect fauna of water-filled tree holes in northern California identified two
species of ceratopogonids, two chironomids, two culicids, one syrphid, one psychodid, and one helodid. The most
common insects encountered were immatures of the western treehole mosquito, Aedes sierrensis. Tree hole surface
area and leaf litter weight were correlated with the number of insect species per tree hole. The emergence periods,
habitat characteristics, and possible food of some of these organisms are described.
INTRODUCTION
have been described ( Jenkins and Carpenter 1946,
Garcia and Ponting 1972, Hawley 1985) and a number
Tree holes filled with rainwater and containing
of investigators have studied its pathogens and parasites
fallen leavesand stemsusually form habitatssupporting (
Sanders 1972, Clark and Brandl 1976, Samson and
detritus- based invertebrate communities.Many
ofSoares 1984, Egerter and Anderson 1985, Washburn et
theseinvertebratesarefoundinno otherhabitator are al. 1988).
This is the first study concerned with
largely
restricted to water-filled tree holes (Kitching
examining the other dendrolimnetobiont community
1971). Rohnert ( 1950) used the term " dendro-members from tree holes in oak woodlands of northern
limnetobiont" todescribesuchspecies. BecausesomeCalifornia. The biology and taxonomy of the insects
oftheseinvertebratesarerarelycollectedorstudied,the associated with most other culicid vectors of disease are
systematics, especially of the immatures, are often
well known when compared to those associated withAe.
poorly known. As a result, studies of tree-holesierrensis. A more thorough understanding of the entire
communities have rarely includeda complete species
tree-hole community may be beneficial in control
list( Fish1983). The only known North American study efforts against this mosquito.with a complete list was provided by Snow( 1958) in his
analysis of the spatial distribution of the insect
MATERIALS AND METHODS
community in a water-filled stump in Illinois. Rohnert
1950)and
Kitching(
1971) providedEuropeanspeciesNorthern oak woodlands ( Munz 1965) of Lake
lists from water-filled tree holes in
Germany
andCounty and eastern Mendocino County ( Longitude
England, respectively.Kitching
andCallaghan(1981)122° 38 IV to 123° 06' W, latitude 38° 42' N to 39° 14' N)
reported a specieslist forwater-filledtreeholes ina rainwere searched for water-filled tree holes. Most large
forestofsouthernAustralia.Kitching(
1983)comparednative trees in these areas are Quercus lobata Nee
the communitystructure of water-filled tree holes in (
valley oak), Quercus kelloggii Newberry ( California
EuropeandAustralia.
black oak), Quercus wislizenii Candolle ( interior live
Water- filled tree holes in oak woodlands of theoak), and Arbutus menziesii Pursh( Pacific madrone).
northernCalifornia Coast Range have beenstudied
by
Elevation in the study area ranges from ca. 365 to 425 m
anumber of workers sincelarvaeofthemosquitoAedes
above sea level. Rainfall averages 71 cm annually in the
sierrenis(Ludlow)arethemost commoninsectsinthat
city of Lakeport, near the center of the study area.
habitat. Femalesofthisspecies are serious
biting
pestsSummers are hot and dry. Winters are wet and mild with
of man anddomesticatedanimalsinmanyareas ofthemost rain falling in November to March of each year.
Pacific Northwest( Bohartand Washino1978,Papineau
Based on a yearly cycle of July 1 to June 30, the
1983-1984)andhave been implicatedasimportantvectorsof
84 and 1984- 85 seasons produced 93. 7 and 53. 2 cm of
Dirofilaria immitis(
Leidy),
thenematode responsiblerainfall, respectively( Fig. 1).
for
dog
heartworm disease ( Weinmann and GarciaTwo
types of water-filled
tree holes were
1974). The life
history
andbiology
ofthis mosquitoidentified. The most common occurred with regrowth
222 BULL.SOC. VECTOR ECOL. DECEMBER, 1988 40 35 MIN. TEMP. MAX. TEMP. 35— 30••: •, i. RAINFALL , S 30 —
II
t % % Ii-
25 —II
20 V
FW--
20 — t / la..P
Z• 15S
Z j 0 10 I e5-1
p
1
i /.
o ;. !
00 .
rp o J A S O N D J F M A M J J A S O N D J F M A M J1983
1984
1985
Figure 1.
Mean monthly high and low temperatures and monthly precipitation near Lakeport, Lake County,
California from July 1, 1983 to June 30, 1985. ( Precipitation data were provided by the Lake County
Air Quality Management District, Lakeport, CA. Temperature data were supplied by Albert Reuss,
DECEMBER, 1988 BULL. SOC. VECTOR ECOL. 223
of multiple stems
following
cutting or death of thewere teased apart in a droplet of deionized water on a
originalprimarystem. Afteryears of regrowth,apan, microscope slide. The slide was viewed at 40- 1000Xoften capableof
holding
rainwater,occursinthecenterunder a compound microscope for identification of the
ofaringofsecondarytree trunks.Panswerefoundfrom gut contents.
ca.0. 2mbelowto1. 0mabove groundlevel. Tree holes
The predatory capacity of Culicoides spp. larvae
ofthisformwere reportedtobethemostcommontype
for Ae. sierrensis larvae was tested in the laboratory. A
inan oaksavannahin Wisconsin
by
HansonandHansonprevious study ( Clark and Fukuda 1967) reported
1970). Rot holes formafterabranch breaksoff of apredation by larvae of Culicoides cavaticus Wirth and
tree. The remaining portion and the adjoining trunk
Jones upon fourth instar Ae. sierrensis. A total of 10
may become hollow and hold rainwater. Rot holes
replicates was done to analyze the extent of this
sampled were ca. 1.5to6. 0mabove ground.Kitching
phenomenon. Seven replicates tested fourth instar Ae.
1971) defined "pans" and " rot holes" in a similar
sierrensis. Three replicates were completed using first
manner.
through third instar Ae. sierrensis in approximately
Pans in 32
Q.
kelloggii, 4Q.
wislizenii, 3 A.equal numbers in each replicate. Ten ml of filtered
tree-menziesii,and5rotholesinQ.
lobataweresurveyedforhole water( 801.1. filtration) was added to each of 20, 50
immature insects from 1983through 1985. Thesetreeml glass beakers. Ten Ae. sierrensis larvae( instars as
holes arelocatedat eight sites between Lower Lake, described above) were added to each beaker. Ten Lake
County,
and Lake Mendocino in Mendocinofourth-stage larvae of Culicoides spp.( collected from 5
County.
The treeholes are separatedby
a maximum tree holes) were added to each of 10 of the beakers. Thedistanceof56km. other 10 beakers served as controls. Each beaker
Qualitative sampling for insectswas accomplished
containing both genera was observed at irregular
by
stirringthecontentsofthe treeholeandremovingaintervals for visual evidence of predation. The number
150mlsampleof wateranddetrituswitha polyethylene of Ae. sierrensis larvae alive in each beaker was pipette.Themouth ofthepipettewas5mmin diameter, determined after 40 hours.largeenoughto collect maturelarvaeofeachdendro- In the last week of March, 1985, a water sample
limnetobiont insectspecies. Insectswereidentifiedand was collected from 12 of the 39 pans surveyed for countedinenamelpansinthefieldand returnedto the
immature insects. These included five Q. kelloggii,
treehole. Thisprocedurewasrepeatedthree timesforfour Q. wislizenii, and three A. menziesii tree holes. A
eachtreeholepersampling date. Eachtreeholewas
similar sample was collected from three Q. lobata pans
surveyedfrom3 to24timesbetweenNovember 7and in the study area.
The pH and conductivity were
May
3of1983- 84and1984-85. Fifteenofthe treeholesmeasured by electrode in the laboratory. All leaf litter
weresampled on21 dates in 1983- 84toinsurethatno was removed from each pan, drained, weighed, and species was missedbecauseofimpropertimingofthe returned to the tree hole. Emergence traps were then
surveys. Theothertreeholesweresampled on3to 10 placed over each of the 15 pans. Emergence traps were
dates each. Mean length andwidth of eachtreehole
constructed of surgical stocking painted black on the
were determined
by
averaging a series of measure- outside. One end was stapled and taped over the tree-ments. Theproduct of meanlengthand meanwidthwashole opening. The other end was attached to an
used toestimate surface areaofeachtreehole. Max-inverted funnel. A 250 ml clear plastic cup was placed
imumdepthof eachtree holewhenfull ofwaterwasover the small opening of the funnel so adult insects
recorded.
could enter the container. Each trap was maintained in
Except forthechironomidLimnophyes hamiltoni
a taut, upright position by wires connected to branches
Saether, larvae of all species presumed to be and trunks of the tree. Adult insects which did not dendrolimnetobionts were reared to adults in thefly into the cup were aspirated out of the surgical
laboratory.
Larvae ofL. hamiltoni are difficult tostocking through a zippered opening. Emerging adults
maintain in thelaboratory,
but field-collected pupae were collected from each tree hole once per weekwere reared to adults. Culicids were identified
beginning
March 25, 1985. Collections were according to Bohart and Washino ( 1978). Adultsofterminated when the tree holes dried completely and
other species were identifiedby
several taxonomists adult emergence stopped. Some larger or deeper treeseeAcknowledgements). holes were monitored until September 2, 1985. After Gut-content analyseswereperformedon all ofthe emergence data were collected for the season, the non- culicidtree-hole insects.
Freshly
field-collectedvolume of each tree hole was determined by emptying
larvae were washed and the digestive tracts werethe hole and filling it to capacity with a measured
224 BULL.SOC. VECTOR ECOL. DECEMBER, 1988
RESULTS in tree holes in the eastern United States, uses a cibarial filter to feed upon fine-grained tree hole detritus. The
The 39pans surveyedhadameansurface area of digestive tracts of 12 B. humeralis larvae examined 335. 7cm2(
range36-1064cm2).
The meanmaximum
during this study contained primarily ciliate protozoans
depthmeasured 10.9cm( range4- 29cm). The fiveQ.
and fine-particle detritus.lobatarotholes hadameansurface area of1499cm2
One B. humeralis pupa was collected in moist leaf
range 25- 4800 cm2)
and amean maximum depthof
litter above the waterline in a tree hole. A
laboratory-57. 0cm(range22- 171 cm). reared adult emerged after 15 days in the pupal stage. Nine species of insects werefrequently
Adults were collected in tree hole emergence traps from encountered in the 44 tree holes surveyed and are the end of April until earlyJuly,
1985. Emergence included inthelistofdendrolimnetobiontinsects from trapping showed a rather synchronous emergence the study area (TABLE 1). A description of each pattern in each tree hole with a single collectivefollows. emergence peak(
Fig.
2A). Mallota posticata, anothertree- hole inhabiting syrphid is known to be univoltine
Blerahumeralis Maier 1978).
The adults of this syrphid have been collected in
Washington, Idaho, Oregon,andCalifornia( Cole 1969) Limnophyes hamiltoni
and werefirstdescribed
by
Williston(1882).AdultsareAdults of this midge were originally described by
brown with numerous yellow areas that include the
Saether( 1969). They are ca. 2 mm in total body length.
humeriandbasal halvesofthe femora. Although theThe Lake County records from this study represent a
adults weredescribedover100yearsago,priorto this considerable extension in the known range of the studytheimmaturesand theirlife
history
were unknownspecies. Saether( 1984) recently reported collection of
F. C.Thompson,pers.commun.). L. hamiltoni in South Carolina. Prior records are from Larvae were collected inatruly aquaticsituation Greenland and Alaska as well as the Northwest and
in freewaterorin detritusbeneathfreewater)inthe tree Yukon Territories and British Columbia, Canada. In
holes. Themature larvaeare 15-18 mm
long
with aaddition, L. hamiltoni is probably a synonym of a
breathing
tube thatcan beextendedtoover60mminPaleartic species with several earlier names including
length. Maier (1978) reportedthatMallotaposticata Limnophyes smolandicus and Limnophyes vernalis Fabricius),anothersyrphid
fly
whoseimmaturesoccur ( Brundin 1947). Despite the extensive geographicTABLE 1. Dendrolimnetobiont insects collected from water- filled tree holes in oak woodlands in northern California.
Order
Family
Genus SpeciesDiptera Syrphidae Blera humeralis( Williston)
Chironomidae Limnophyes hamiltoni Saether
Polypedilum pedatum excelsius Townes
Ceratopogonidae CulicoidesneofagineusWirth& Blanton
CulicoidescavaticusWirth& Jones
Culicidae Aedes sierrensis( Ludlow)
Orthopodomyia signifera( Coquillett)
Psychodidae
Telmatoscopus sp.( undescribed)
DECEMBER, 1988 BULL. SOC. VECTOR ECOL. 225 12 A
1
10 ELENA a 8 CYPNON1
J cs u. e1
0ac
US 4 211
O 60 46he
US 40
W3
36 POL YPEO/L UMW
CL
07 30 L/ MNOPNYES J o= 26au-
20m15
10 j/ \ 5 0 30 C. C4VA77CUS C 25 C. NEOPAC/NEUS CE a 20 - 7ZZA447t7SCCCPUS 1•J 16 -tU-0
10W
m
z
6 lMAR APR MAY JUN JUL AUG
Figure 2.
Numbers of adult dendrolimnetobiont insects( excluding Culicidae) caught in emergence traps over
15 pans in northern California oak woodlands from March 28 to September 2, 1985. A) Blerahumeralis and Cyphon sp., B) Polypedilumpedatum ecelsius and Limnophyes hamiltoni, C) Culicoides
cavaticus,Culicoides neofagineus, andTelmatoscopus sp.( threepointrunningmeans).226 BULL.SOC.VECTOR ECOL. DECEMBER, 1988
range known for this species, all of the previous water-filled tree holes. Each of these holes contained
collections wereofadults, thelarvaeandtheirhabitat
Limnophyes larvae. Sweep net collections were made
being
unknown (D. R. Oliver, pers. commun.).from one of those swarms. Forty-six males and eight
Grodhaus and Rotramel ( 1980) previously reportedfemales were collected and counted in the laboratory.
collection of Limnophyes larvae from northern
Adult production of L. hamiltoni from overwintering
California tree holes but they did not
identify
the larval populations in trapped tree holes ceased inmid-species.
June 1985( Fig. 2B), earlier than any other of the
tree-Withintheconfines ofthestudyareas examined,L.
hole inhabiting insects studied.
hamiltoni immatures are apparently largely restricted to
water-filled tree holes. Other species in the genus Polypedilum pedatum excelsius
inhabitthemarginsandshallowregions ofalltypesof Polypedilum pedatum excelsius and Polypedilum
fresh waterand somelive in
damp
terrestrialhabitats pedatum pedatum are the western and eastern NorthOliverandRoussel 1983). Searchesof
damp
terrestrialAmerican forms, respectively, of the same species
habitatsand moistleaf litterinthestudyareasdidnot ( Townes 1945). Polypedilum pedatum excelsius hasresultin locationofLimnophyes larvaeoutside of water- been collected from Washington and California( Cole
filledtree holes. Larvae were common inpans of
Q.
1969). Grodhaus and Rotramel( 1980) described all thekelloggii,
Q.
wislizenii, andA. menziesiibutwerenot immature life stages and reported larval habitats and lifefoundin
Q.
lobatarotholes( TABLE2). Noadultswerehistory of the western subspecies. All of their records of
collectedfromQ.
lobatapans( TABLE 3).larval habitats are from oak ( Q. kelloggii, Q. lobata,
Limnophyes larvae are sprawlers ( Merritt and Quercus agrifolia [Neel]) and laurel ( Umbellularia Cummins 1984) andwere most commonintreeholes californica[ Hooker and Amott]) tree holes except for a that werenearly filled withleaves anddebris. Larval single collection from wet leaf litter at the edge of a gut-content analyses(n=21)showed thedigestivetracts spring under oak and laurel trees. Larvae of most
containeddetritusparticlesandtoalesserextentfungal species in the genus live in bottom substrate, often hyphae.Pupaewerefoundatthewatersurfaceofsome associated with plant debris( Oliver and Roussel 1983).
treeholes. Neither larvaenor pupae wereeverfound in
In the present study, larvae were most frequently
tubesor cocoons. collected from A. menziesii tree holes ( TABLE 2).
Swarms ofadultL. hamiltoni were observed on
Adults were collected only from pans of A. menziesii
February
27, 1985,circlingabovetheopeningsoftwo ( TABLE 3).TABLE 2. Percentages of tree holes positive for immature dendrolimnetobiont insects in the various tree species
of oak woodlands in northern California in 1983- 85.
Pans Rot holes
Quercus Quercus Arbutus Quercus All
Organism kelloggii(32)* wislizenii(4) menziesii(3) lobata(5) tree holes( 44)
Aedessierrensis 100.0 100. 0 100.0 100.0 100.0
Orthopodomyiasignifera 3.1 0. 0 0. 0 60.0 9. 1
Culicoides spp. 96.9 100. 0 100.0 100.0 97. 7
Blera humeralis 15. 6 75. 0 66.7 40.0 27. 3
Polypedilumpedatum excelsius 3.1 25. 0 66.7 0. 0 9. 1
Limnophyes hamiltoni 46.9 75.0 100.0 0.0 47.7
Telmatoscopus sp. 37.5 75. 0 66.7 0.0 38. 6
Cyphon sp. 12.5 0.0 0.0 60.0 15. 9
DECEMBER, 1988 BULL. SOC. VECTOR ECOL. 227
TABLE 3. Numbers of adult insects collected from emergence traps over 15 pans from March 28 to September
2, 1985. y h c p, byP;
Arbutusmenziesii 1 1331 0 17 3 295 2 5 0 2 10 0 8 49 67 3 5 0 3 13 0 16 4 0 1 17 0 Quercuswislizenii 1 4 0 3 1 0 2 0 0 2 102 3 29 6 0 10 0 0 3 1403 2 25 2 0 0 0 0 4 201 1 89 0 0 0 11 0 Quercus kelloggii 1 16 16 0 1 0 0 9 12 2 193 10 20 0 0 6 15 0 3 527 23 25 0 0 0 14 55 4 28 0 58 0 0 1 4 0 5 101 0 2 2 0 0 0 0 Quercus lobata 1 452 0 0 0 0 0 0 0 2 7 0 26 0 0 0 0 0 3 14 0 0 0 0 0 1 0Alltreespecies x 293. 5 3. 7 21.2 4.5 24. 1 1.7 5.4 4. 5
Live fourthinstar P. p.excelsiuslarvaeareblood
inhabiting culicids in the study area have been
redincolor. Larvaeconstructtubesonleavesand otherextensively studied. A few of the important Ae.
treehole debrisandwereveryseldom collected outside sierrenis studies are cited in the introduction. Chapman of them. Near the end of summer,live larvae canbe (
1964) and Zavortink( 1985) have studied the biology
collectedfromsometreeholeswhichhave lacked freeand ecology of Or. signifera. The range ofAe. sierrensis
waterfor overtwo months (Grodhaus and Rotramel includes much of western North America from British
1980).
During
the wet season, larval gut-contentColumbia to Baja California. Orthopodomyia signifera
analyses(n=11) showedthedigestivetracts tocontainhas been found in most of temperate North America.
flagellate protozoans, fungal hyphae, and detritusBoth are largely restricted to tree holes ( Bohan and
particles. Washino 1978). Tree hole Aedes larvae in the
Adultswerecapturedintree-holeemergencetraps
southwestern United States feed by filtering, browsing,
from the
beginning
of April into late August, 1985. and gnawing ( Zavortink 1985). OrthopodomyiaThere were two early season emergence peaks, the signifera larvae are filterfeeders- detritivores( Lounibos
largest occurringneartheend of
May( Fig.
2B). A later 1985).season emergence peak coincided with
drying
oftheAedes sierrensis and Or. signifera larvae usually
treeholes.occur together only in permanent rain-filled tree holes.
Orthopodomyia signifera dominates permanent tree Aedessierrensis andOrthopodomyiasignifera
holes filled with fluids secreted by the plant( e.g., some
Thebiology
and ecology of the two tree hole willowsand cottonwoods) andAe. sierrensisdominates228 BULL. SOC. VECTOR ECOL. DECEMBER, 1988
temporaryrain-filledtreeholes( Zavortink 1985). Each
Culicoides cavaticus and Culicoides neofagineus
oftherotholecollections ofOr.signiferainthisstudy
The larvae of these two species are undescribed
occurredin
deep
holeswithrestricted openingswhichand indistinguishable from each other ( W. Wirth, pers.
allowedrainwatertoremainintheholethrough atleast commun.).
Adults are markedly different in appearance
mostofthedry
summerperiod.Water inthese treeholesand easily distinguished by using the descriptions of
is of high pH (mean=8.12) and conductivityWirth and Jones( 1956) for C. cavaticus and Wirth and
mean=2937 µmhos/cm) and very dark in color. Blanton ( 1969) for C. neofagineus. Larvae of each Nonetheless, the Or. signifera larvae collected from
species have only been reported from tree hole habitats.
these holes were pink in color
indicating
they hadThe feeding habits of adult females of each species are
ingestedthephotosyntheticbacteriaThiocapsa( Bohartpoorly known but C. neofagineus has been collected
andWashino 1978). In onlyonetreehole,aQ.
lobata from the ears of deer and in quail- baited traps( Wirth and rotholewithadepthof171cm,did Or.signiferalarvae Blanton 1969).The known range of each species
outnumberAe.sierrensislarvae. includes parts of Arizona, California, and OregonAedes sierrensis adults were collected in ( Wirth et al. 1985). Emergence traps were placed on 15 emergence trapsfrom the first week of April to the
pans in 1985 and 13 produced Culicoides adults. Five
secondweekofAugust, 1985. Maleemergence showed produced adults of both species, seven produced C. astrongpeakin April. Female emergencewasmore
cavaticus only, and one produced C. neofagineus only
evenfrom AprilthroughJune,thendeclinedand ended ( TABLE 3).
inmid-August(
Fig.
3). Maleadults were59. 9percent In 1985, adults of each species emerged into tree-of thetotalemergencefromthe15panstrappedoverthe hole traps in the first week of April. Culicoidescavati-entire 1985season. Notrapswereemployed overtree
cus showed strong emergence peaks in early spring and
holescontaining Or.signiferalarvae in 1985, butinaas the tree holes dried near the end of July. Culicoides
separatestudyin 1986,oneA.menziesiirotholewitha neofagineus showed similar but less marked
emer-smallpopulationofOr.signiferalarvaewasmonitored gence peaks.
Emergence occurred from early April
with anemergencetrap. Adultsemergedprimarilyin
until the tree holes dried at the end of August ( Fig.
themonths ofSeptemberandOctober. 2C).900 SO-MALES 700- FEMALES W 600-3 600-4 400-O 1
52
r ZooI \\
100- V oMAR APR MAY JUN JUL AUG
Figure 3. Numbers of male and female Aedes sierrensis caught in emergence traps over 15 pans in oak
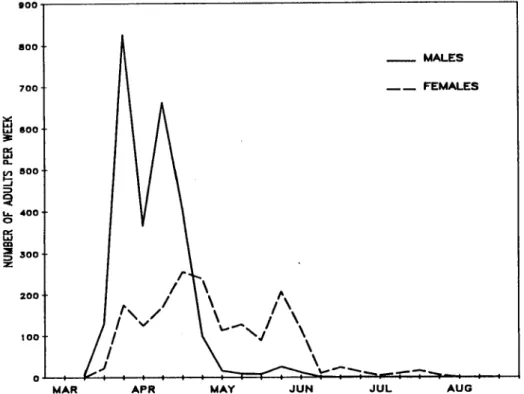
DECEMBER, 1988 BULL. SOC.VECTOR ECOL. 229
ClarkandFukuda(1967)reported predation
by
C.in the laboratory were found to contain large particle
cavaticus larvae upon Ae. sierrensis larvae. Their
tree hole detritus, flagellate protozoans, and fungi.
observationsweremadeinthe
laboratory
inwaterfreeAdults emerged into tree hole traps from early April
from any debris,and whenalfalfa pellets were added, until September in 1985 (Fig.
2C). The emergencepredation wasno longer observed. Hair and Turner
pattern of overwintering larvae is similar to that of the
1966) reported that larvae of Culicoidesguttipennis
Culicoides spp. and shows small spring and late summer
Coquillett), a treehole speciesin theeasternUnited emergence peaks.States, sometimes feed upon live Aedes triseriatus
These flies are easy to maintain in the laboratory. A
Say),
Or.signifera,andhelodid larvae.self-perpetuating colony can be maintained by placing
The digestive tracts of Culicoides spp. larvaelarvae, tree hole water, and leaves in a petri dish in a
n=21)collectedinthestudyareawerefoundtocontainsmall cage. Females lay cream colored cylindrical
fineparticledetritus, flagellateprotozoans, and smaller eggs on leaves on the surface of the water which hatchvolumes offungalparts. Thegutsofsomelarvaealso
within two days. No evidence of delayed hatching or
contained afewalgalcells. Inthe
laboratory,
predationdesiccation- resistant( aestivating) eggs was found. One
wasnot observeduponany larval instar in beakers,each
liter of airspace is sufficient to maintain mating and
containing 10 Culicoides larvaeand 10 Ae.sierrensis
oviposition. In the absence of other tree hole species,
larvae. After48 hours, Ae.sierrensislarvalmortalitylarval density often became very heavy in the laboratory
averaged7. 0percentinthe 10replicatesthatincludedcolony but was always relatively sparse in the tree holes
Culicoides larvae. Control mortalityduring
thesame surveyed.period averaged 5. 0 percent. There was no significant difference (Student's t-test, P>0.05) in Ae. sierrensis
Cyphon sp.( undescribed)
mortalitybetween beakerscontaining Culicoides larvae
This beetle is the only non- dipteran insect among
andcontrolreplicates. Culicoides larvaeusedforgut-the dendrolimnetobiont fauna in the study area. The
content analyses andlaboratory
bioassays were range of this undescribed species isunknown. In collectedfromtree ho!eswhichproduced adults ofC.
addition to the Lake County records from this study,
cavaticusonly, C.neofagineusonly,andbothspecies. there is a single adult specimen from Crater Lake
National Park, Oregon in the collection of the U. S.
Telmatoscopussp.( undescribed)
National Museum. Based on comparisons of the adult
Thegeographic rangeofthisundescribedfly
isnot genitalia and the presence of subbasal pronotal known outside of Lake and Mendocino Counties. depressions, this undescribed species is thought to beAdults have beenexamined
by
Dr. Derek Duckhouse,closely related to the eastern North American species,
whoplacesthespeciesinthe genusClogmia. Quateand Cyphon americana Pic and Cyphon cooperi Schaeffer. Quate ( 1967) recognized Clogmia as a subgenus of
Females can be separated from described members of
Telmatoscopus. At that time, Duckhouse alsothe genus by the presence of two contiguous pits on the
recognized Clogmiaatthe subgeneric level, but laterlast ventral abdominal segment, each containing
dark-Duckhouse( 1974) elevatedittofullgenus. Quateandpigmented sensory hairs. A description of this taxon is
Vockeroth(1981) stillhadnot recognized genericlevel in preparation( G. Kennen, pers. commun.).for Clogmia, so the
fly
is listed hereunderthename Larvae are crawlers and can be collected on the more widely used in North America. Most of the walls of the tree holes as well as in detritus at the bottom.Clogmia spp.areAustralianandAsian but inadditionto
In the laboratory, larvae pupate in leaf litter or crevices
this undescribed species, macdonaldi Quate, in bark above the water line. Larvae of all instars can be albipunctata Williston, and superbus Banks have collected from moist detritus in the bottom of tree holesNearctic distributions. The species collected in this with no free water at the end of summer. Pupae have
study is closelyrelatedtomacdonaldibut apparently is
four long spines on the pronotum similar to those
not conspecific. Duckhouse ( 1978) reportedthat the reported for Prionocyphon limbatus LeConte ( Leech larvae ofClogmia spp. occur mostly in treeholes or andChandler 1956). In 1985, adult emergence did not
plant containers. This undescribed species was
commence until the tree holes began to dry up.
collectedfrompans of allfourtreespecies examinedbut
Emergence of adults peaked at the end of July( Fig. 2A).
wasabsentin
Q.
lobatarotholes( TABLES 2and3). The single emergence peak, coupled with observations The larvaeofthis psychodid areburrowers intheduring the study that all larval instars are present in tree
detritus intreeholes.
They
arefoundmostcommonlyholes during all of the yearly seasons, suggest that this
inshallowtreeholeswithlargeamountsofleaf litter.species may spend more than one year in the larval
The digestivetractsof14larvae dissectedand analyzed stages.
230 BULL. SOC. VECTOR ECOL. DECEMBER, 1988
morethan oneyearis implied foraEuropean helodid
contained depressions or ledges above the waterline
Prionocyphon serricornis Muller). He found thatwhich contained moist leaf litter. Insects occasionally
larvaldensities were similar allyear intreeholes andcollected from tree holes during this study which are not
thatlarvae live upto twoyearsinthe
laboratory.
dendrolimnetobionts included Culiseta incidens
Peterson ( 1960) stated that helodid larvae are (
Culicidae), Tipula( Tipulidae), Rhagio( Rhagionidae),
predaceousbut other authors (Kitching
1971, Merritt Dasyhelea ( Ceratopogonidae), and Empididae. and Cummins 1984) refer to themas saprophages orCollembolans, including Sminthurides( Sminthuridae)
herbivores. Gut-content analyses of larvae of thisand Isotomidae, were also present in some tree holes.
Cyphon sp. (n=18) showed the larval diet to consist
Physical and water quality parameters of 15 pans
mainly of detritus particles and fungi. A smallused for analyses of adult insect production over an
percentage ofthedietconsisted offlagellateprotozoans entire season are presented in TABLE 4. Quercusand, in one treehole, submergedlichens. Larvaeare
lobata pans had relatively small mean surface area, low
easily reared to adults in thelaboratory
without themean leaf litter weight, high mean pH, and high
presence ofpotentialpreyspecies. conductivity compared to the other tree species
measured. Quercus wislizenii pans had relatively low
DISCUSSION
mean pH and conductivity and were the shallowest pans
studied. Tree holes with high pH and conductivity had
Although all the insect surveys were qualitative, nearly opaque coffee- colored water. Water in acidic Ae. sierrensiswas the most prevalent andCulicoides
tree holes was transparent with a light brown tinge.
spp. were the second most encountered immatureEmergence trap data from each of the 15 pans are
insects inthesurveys. Thesetwogenera were alsothepresented in TABLE 3. These collections demonstrate
most prevalentintermsof numbers oftreeholespositive
the dominance of Ae. sierrensis in terms of numbers of
for their presence (TABLE2). Culicoides spp. wereadult insects per tree hole. Each tree hole produced an
found in 43 of the 44 tree holes surveyed andAe.average of nearly 300 Ae. sierrensis adults, over 81
sierrensis wasfound inallofthem. Thechironomids percent of the total number of adult dendrolimnetobiontand psychodids were absent from allof therotholes insects produced
by
the tree holes. Garcia andsampled. Allwere more prevalentintheshallower pans
DesRochers( 1976) reported that the numbers of adult
whichusually hadleaf litternearlyto thesurface ofthe
Ae. sierrensis produced by a tree hole may be as little as
water. Polypedilum pedatumexcelsius was therarer2.5 percent of the first instar larvae which initially
chironomid but all of the tree holes positive for itshatched in that hole. The data in TABLE 3 do not,
presence also containedL. hamiltoni. The
frequency
ofhowever, indicate the potential total seasonal output of
occurrence of larvae was significantly higher in the
adult insects from the trapped tree holes since the traps
deeper rot holes than in pans for both Cyphon (chi-
prevented oviposition during the season by any species
square P< 0. 01) and Or. signifera (P< 0. 001). which are multivoltive. Also, some emergence of L. Orthopodomyia signiferausually occurs only in treehamiltoni occurred before traps were placed over the
holes that hold water all year (Bohan and Washino tree hole openings.
1978). Cyphonadults emergedvery late intheseason
Each of the 15 pans produced an average of 4. 1
after most oftheshallowtreeholesweredry.
Thereisdendrolimnetobiont insect species with a range of 1 to
also evidencethatsomehelodidsrequire two yearsin6 species per tree hole. Surface area of the tree hole and
the larval stage (
Kitching
1971). Blera humeralisleaf litter weight were both positively correlated with
occurredwithsimilar
frequency
inbothpans and rot the number of species per tree hole( TABLE 5). Surface holes.area is probably the most important physical factor
In addition to dendrolimnetobionts, Rohnertmeasured since it may influence other factors. Surface
1950) and
Kitching (
1971) have described otherarea is positively correlated with leaf litter weight
classifications ofinsectssometimes collectedinwater- (linear correlation coefficient=0. 89, Student' s t test,
filledtreeholes. These includegeneralists attractedto P<0. 05).
Large surface area allows more leaf litter to
a wide range ofhabitatsand not
largely
restrictedto treeaccumulate in a tree hole, which increases the nutrient
holes and otherinsects that occasionally occupy tree
base of the community and provides a habitat for the
holes
by
accident. Snow ( 1958) analyzedthe spatialsprawlers, burrowers, crawlers, and tube builders.
distributionofinsects inawater-filledtreeholeandinLarge surface area also allows more dilution of water in
addition to theaquatichabitat describeda numberof
the tree hole with rainwater. Of the physical factors
other
dry,
moist, and semi- aquatichabitats withinthemeasured, only leaf litter weight was significantly
treehole. Thetreeholes examinedinthisstudyoften correlatedwithtotal adultinsectproduction.DECEMBER, 1988 BULL. SOC. VECTOR ECOL.
231
TABLE4.
Mean physical and water quality measurements of 15 pans taken between March 18- 25, 1985 in Lake
and Mendocino Counties, CA ( 3- 10 days before emergence traps were placed over the tree hole
openings).Arbutusmenziesii Quercuswislizenii Quercuskelloggii
Quercus lobata
NumberofTrees 3 4 5 3 Surfacearea( cm2) 390. 70 ( 160- 570)* 150.00 ( 36- 294) 251. 40( 198-525) 47. 00( 24- 81) Maximumdepth(cm) 10.50 ( 7- 12) 7. 00 ( 4- 11) 19. 40 ( 9- 29) 12. 20 ( 8- 20) Maximumvolume(1) 4. 70 ( 0. 7- 10. 2) 1.60 ( 0.4- 4.6) 8. 70 (1.0- 24.6) 3. 10 ( 0.7- 5. 6) Leaf litterweight(g) 1003. 30 ( 100- 1570) 370. 00 ( 100-950) 534. 00 (105-865) 40.70 ( 10- 62)
pH 6.82 (6. 78- 6.87) 6.31 ( 5.94- 6.74) 7. 15 ( 6. 82- 7. 65) 8. 10 ( 7. 61- 8.48)
Conductivity(µ
mhos/ cm) 566 ( 413- 832) 424 ( 187-913) 905 ( 183- 2041) 3014 ( 1492- 4780)Numbers in parentheses are the range of the parameter measured.
Snow( 1958)studied awater-filled stump in Illinois
have been reported in the tree hole habitat. The absence
andproducedtheonlyprevious specieslistoftreehole
of a predatory culicid in northern California may be
insectfauna fromtemperateNorth America. The onlyrelated to the hypothesis that fewer trophic levels may
species common tohis listand later identified inthis
occur in tree hole community systems which experience
study is Or. signifera. A predatoryculicid,Toxoryn-
more environmental perturbations ( Kitching 1983).
chites rutilus septentrionalis(DyarandKnab),wasalsoRegions where rainfall, desiccation, and leaf fall are less
listedby
Snow. The Illinoistreeholecommunitywasseasonal may support more trophic levels than occur in
similar to that ofnorthern California in
having
one northern California tree holes.detritivorousaedine mosquito(Aedestriseriatus),one
The nine species of insects in the northern
syrphid (Meromarcus acutus [Fabricius]), twoCalifornia tree holes studied are all primarily
ceratopogonids ( Culicoides guttipennis [ Coquillett]
detritivores or saprophages. To the limited extent that
andCulicoidesarboricolaRootandHoffman),and onesome species feed upon photosynthetic bacteria or
helodid (Prionocyphon discoides [Say]).
The majoralgae, some energy utilized by the dendrolimnetobiont
differences between the two tree hole insectinsect community is not detritus based. The literature
communities are the presence of two chironomidindicates that C. cavaticus can be an occasional predator
species in northern California and the presence of abut gut analyses and preliminary laboratory
predatoryculicidin Illinois. NorthernCaliforniaisthe
experiments did not indicate that any of the associated
only area ofNorth America fromwhichchironomidsspecies consume many Ae. sierrensis larvae. However,
TABLE 5.
Spearman's correlation coefficient( rho) for physical factors affecting number of dendrolimnetobiont
species and adult insect production from 15 pans trapped from March 28 to September 2, 1985.
Surface Tree hole Tree hole Leaf
Numberper Area Depth Volume Litter
Conductivity
Tree Hole cm2) cm) cm3) wt.(g) pH (µ mhos/ cm) NumberofSpecies 0. 7767* 0. 1170 0.1661 0. 7750* - 0. 3866 - 0. 2393 NumberofAdults 0. 3848 0. 0098 0. 1643 0.5384* - 0.3866 - 0. 2179 Significant Spearman's rank correlationcoefficient(P< 0. 05).232 BULL. SOC.VECTORECOL. DECEMBER, 1988
most of these cohabiting populations could at times Cole, F. R. 1969. The flies of western North America.
competefor foodor spaceintheserestrictedhabitats. In
Univ. Calif. Press, Berkeley, 693 pp.
a similar tree hole community in beech trees in England
also
lacking
predators),Kitching (
1983) concluded Duckhouse, D. A. 1974. Redescription of thethatthedetritus- basedresourcesare partitioned
by
the neotropical Psychodidae ( Diptera, Nematocera) tree-holeinhabitantslargely
basedontheparticle sizedescribed by Rapp and Curran. J. Entomol. ( B)
utilized as wellas spatial andtemporal means. The 43( 1): 55- 62.
interrelationships of the various tree hole species in
northernCaliforniaareunknownand thecommunity Duckhouse, D. A. 1978. Non- phlebotomine
warrantsfurther study. Psychodidae ( Diptera, Nematocera) of southern
Africa, II. Subfamily Psychodinae: Neoarisemus
Acknowledgements and the brunettoid and telmatoscopoid genera.
Ann. Natal Mus. 23( 2): 305- 359.
The identifications
done by the following
taxonomistswere essentialforthisstudyand aregreatly Egerter, D. E. and J. R. Anderson. 1985. Infection of appreciated: Mr. Gail Grodhaus, California the western tree hole mosquito, Aedes sierrensis DepartmentofHealthServices,
Berkeley,(
Culicoides Diptera: Culicidae), with Lambornella clarki andPolypedilum),Dr.Charles Hogue, NaturalHistory
Ciliophora: Tetrahymenidae). J. Invert. Pathol. Museum of Los AngelesCounty,
Los Angeles, 46: 296- 304.California( Blera and Telmatoscopus), Dr. Don Oliver,
Biosystematics Research Institute, Agriculture Canada,
Fish, D. 1983. Phytotelmata: flora and fauna. Pp.
1-Ottawa,Canada( Limnophyes), Dr. DerekDuckhouse,
28 in Phytotelmata: Terrestrial Plants as Hosts for
University
of Adelaide, Adelaide, AustraliaAquatic Insect Communities( J. H. Frank and L. P.
Telmatoscopus), Dr. F. Christian Thompson, Lounibos,eds.).Plexus Publishing, Inc., Medford,
Systematic
Entomology
Laboratory,
U. S. D.A.,304 pp.
Washington, D.C., ( Blera),and Mr. Gary Kennen,
Stony
Brook, New York ( Cyphon). InformationGarcia, R. and G. Ponting. 1972. Studies on the ecology
provided
by
Dr.Willis Wirth,SystematicEntomology
of the treehole mosquito Aedes sierrensis (
Lud-Laboratory,
U.S.D. A., Washington, D.C. is also low).Proc. Calif. Mosq. Contr. Assoc. 40: 63- 65.
gratefully acknowledged. We thank Joann Haberthur,
AlbertReuss,andLewisTurlingtonoftheLake
County
Garcia, R. and B. S. DesRochers. 1976. NaturalMosquitoAbatementDistrictfortheircooperation. attrition in larval populations of the treehole mosquitoAedessierrensis(Ludlow). Proc. Calif.
REFERENCES CITED
Mosq. Contr. Assoc. 44: 46.
Bohart, R. M.andR.K. Washino. 1978. Mosquitoesof Grodhaus, G. and G. L. Rotramel. 1980. Immature
California. 3rd Ed., Div. Agri. Sci., Univ. Calif. stages of Polypedilum pedatum excelsius( Diptera, Press,
Berkeley,
153pp.Chironomidae) from seasonally flooded tree holes.
ActaUniversitatis Carolinae- Biologica 1978: 69-Brundin, L. 1947. Zur kenntnis derschwedischen 76.
Chironomoden. Arkiv.Zool.39A(3): 1- 95.
Hair, J. A. and E. C. Turner, Jr.
1966. Laboratory
Chapman,H. C. 1964. Observationsonthebiology
and colonization and mass- production procedures for ecology of Orthopodomyia californica Bohart.Culicoides guttipennis. Mosq. News 26( 3):
429-Mosq.
News24( 4): 432-439. 433.Clark, T. B.andD.G.Brandl. 1976. Observationson Hanson, R. P. and M. G. Hanson. 1970. The effect of
the infection of Aedes sierrensis
by
a land use practices on the vector of California tetrahymenineciliate. J.Invert.Path. 23:341-349. Encephalitis ( LaCrosse) in north central UnitedStates. Mosq. News 30( 2): 215- 221.
Clark, T. B. and T. Fukuda. 1967. Predation of
Culi-coides cavaticus(WirthandJones) larvaeonAedes
Hawley, W. A. 1985. Population dynamics of Aedes
sierrensis(Ludlow).Mosq.
News27( 3):424-425. sierrensis.Pp.
167- 184inEcology
ofMosquitoes:DECEMBER, 1988 BULL. SOC. VECTOR ECOL. 233
Proceedingsof a
Workshop(
L. P. Lounibos, J. R.Papineau, E. A. 1984. Controlling Aedes sierrensis in
Rey,
and J. H. Frank, eds.). Florida Medical JacksonCounty,
a year-round program. Proc.Entomology Laboratory,
Vero Beach, 579 pp.Calif. Mosq. Contr. Assoc. 52: 63- 67.
Jenkins, D. W.andS. J. Carpenter. 1946.
Ecology
ofPeterson, A. 1960. Larvae of Insects: An introduction
the tree hole
breeding
mosquitoes of Nearcticto Nearctic species, Part H: Coleoptera, Diptera,
North America. Ecological Monographs, 16( 1): Neuroptera, Siphonaptera, Mecoptera, 32-47.Trichoptera. 2nd ed., Columbus, 416 pp.
Kitching,
R. L. 1971. Anecological studyofwater-Quate, L. W. and S. H. Quate. 1967. A monograph of
filledtree-holesandtheirpositioninthewoodland Papuan Psychodidae,
including Phlebotomus
ecosystem. J.AnimalEcol. 40: 281- 302. Diptera).
Pacif. Insects Monograph 15: 1- 216.
Kitching,
R.L. 1983.Community
structureinwater-Quate, L. W. and J. R. Vockeroth. 1981. Psychodidae.
filled treeholes in Europe and Australia -
Pp. 293- 300 in Manual of Nearctic Diptera, Vol. 1
comparisons and speculations.Pp.
205- 222 inJ. F. McAlpine, B. V. Peterson, G. E. Shewell, H.
Phytotelmata: Terrestrial Plants as Hosts forJ. Teskey, J. R. Vockeroth, and D. M. Wood, eds.).
AquaticInsect Communities( J. H. FrankandL.P.Agriculture Canada, Ottawa, 674 pp.
Lounibos,eds.).
Plexus Publishing, Inc., Medford,
304 pp.
Rohnert, U. 1950. Wasserfullte Baumhohlen and ihre
Besiedlung.
EinBeitrag
zur FaunaKitching,
R.L.andC. Callaghan. 1981. The faunaof dendrolimnetica. Archiv fiir Hydrobiologie 44:water-filledtreeholes inabox forest insoutheast 472- 516.
Queensland. Australian Entomol. Mag. 8: 61- 70.
Saether, O. A. 1969.
Some Nearctic Podominae,
Leech, H. B. and H. P. Chandler. 1956. AquaticDiamesinae, and Orthocladiinae ( Diptera:
Chir-Coleoptera.Pp.
293- 371 in Aquatic Insects of onomidae). Fish Res. Board Can. Bull. 170: 1- 154.California( R. L. Usinger,ed.). Univ. Calif. Press,
Berkeley,
508 pp. Saether, O. A.1984. Limnophyes er, new species
Diptera: Chironomidae) from Finland with new
Lounibos, L. P. 1985. Interactions
influencing
Nearctic records of previously described species.
production of tree hole mosquitoes in south Entomol. Scand. 15( 4): 540-544.Florida.
Pp. 65- 77 in Ecology of Mosquitoes:
Proceedingsofa
Workshop(
L.P.Lounibos, J. R. Samson, R. A. and G. G. Soares, Jr. 1984.Rey,
and J. H. Frank, eds.). Florida MedicalEntomopathogenic species of the hyphomycete
Entomology Laboratory,
Vero Beach, 579 pp.genus Tolypocladium. J. Invert. Pathol. 43:
133-139.
Maier, C. T. 1978. The immature stages and biology of
Mallotaposticata(Fabricus)( Diptera: Syrphidae). Sanders, R. D.
1972. Microbial mortality factors in
Proc. Entomol. Soc. Wash. 80( 3): 424-440.Aedes sierrensis populations. Proc. Calif. Mosq.
Contr. Assoc. 40: 66- 68.Merritt, R. W. and K. W. Cummins. 1984. An
introduction to the aquatic insects of North
Snow, W. E. 1958. Stratification of arthropods in a wet
America. 2nd Ed., Kendall HuntPublishing
Co.,stump cavity. Ecology 39( 1): 83- 88.
Dubuque, 721 pp.
Townes, H. K., Jr. 1945. The Nearctic species of
Munz, P. A. 1965. A California Flora. Univ.Calif. Tendipedini ( Diptera: Tendipedidae). Am. Midl.
Press,
Berkeley,
1681pp. Nat. 34: 1- 206.Oliver,D.R.andM. E.Roussel. 1983. The insectsand Washburn, J. 0., M. E. Gross, D. R. Mercer, and J. R. arachnids ofCanada, Part11: Thegenera oflarval Anderson. 1988. Predation- induced trophic shift
midges of Canada ( Diptera: Chironomidae). of a free-
living
ciliate: Parasitism of mosquito234 BULL. SOC.VECTOR ECOL. DECEMBER, 1988
Weinmann, C. J. and R. Garcia. 1974. Canine American species of tree- hole Culicoides( Diptera:
heartworm in California, with observations on Heleidae). Proc. Entomol. Soc. Wash. 58( 3):
161-Aedessierrensisasapotential vector.Calif. Vector 168.
Views 21: 45- 50.
Wirth, W. W. and F. S. Blanton. 1969. North America
Williston,S. W. 1882. Contributiontoamonographof Culicoides of the pulicaris group ( Diptera: theNorth American Syrphidae. Am. Phil. Soc. Ceratopogonidae). Fla. Entomol. 52(4):
207-Proc.20: 299- 332. 243.
Wirth,W.W.,A.L. Dyce,andB.V.Peterson. 1985.An
Zavortink, T. J. 1985. Observations on the ecology of
atlasofwingphotographs, withasummaryofthe treeholes and treehole mosquitoes in the
numerical characters of the Nearctic species of southwestern United States.
Pp. 473- 487 in
Culicoides ( Diptera: Ceratopogonidae). Contr.Ecology
of Mosquitoes: Proceedings of aAm. Entomol.Inst. 22( 4): 1- 46.
Workshop( L. P. Lounibos and J. H. Frank, eds.).
Florida Medical Entomology Laboratory, Vero
Wirth, W. W.andR.H. Jones. 1956. ThreenewNorth Beach,579 pp.BULL. SOC. VECTOR ECOL.,13( 2): 235- 242 DECEMBER, 1988
POPULATION TRENDS AND BEHAVIORAL ATTRIBUTES
OF ADULT MOSQUITOES ASSOCIATED WITH DAIRIES
IN SOUTHERN CALIFORNIA
E. T. Schreiber', M. S. Mulls' and J. D. Chaney'
ABSTRACT:
Studies relating to adult population trends and host- seeking activities were initiated at a dairy
operation near Norco, Riverside County, California. The most prevalent species caught in dry ice surveillance traps
was Culex quinquefasciatus Say, followed by Culex erythrothorax Dyar and Culex tarsalis Coquillett. Species of
lesser importance were Culex peas Speiser, Anophelesfreeborn Aitken, and Culiseta particeps( Adams). Habitat
preferences were noted with respect to the species. However, no single one or two habitats were good indicators of
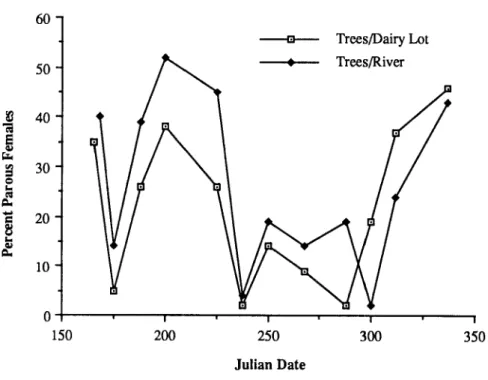
population trends through the season. The peak of host- seeking activity occurred one hour after sunset. Differences
were noted in the peak biting activity of individual species through most of the season. The majority of mosquitoes
captured were nulliparous, yet no differences in host-seeking behavior were noted with respect to parity throughout
nighttime activity.INTRODUCTION these ponds often have low potential for mosquito development( M. S. Dhillon, pers. commun.). Ecological and behavioral information on adult
Dairy operations with poor lagoon management, in
CulextarsalisCoquillett, CulexquinquefasciatusSay,
addition to providing ideal development sites, have a
and Culexpeus Speiser is pivotal in elucidating thecopious supply of blood for host- seeking mosquitoes
epidemiology of St. Louis encephalitis and western
since the cattle are nearby. Effects on milk production
equine encephalitis(BohartandWashino1978)andfor
may occur if heavy populations exist. In Louisiana,
developing
practical controlmethodologiesinsouthernSteelman et al.( 1972, 1973) found weight reduction in
California.
Additionally,
characteristics ofthe adult levels of Hereford and Brahman breed steers due to ecology and behaviorofthe various species mustbemosquito feeding.
understoodtoaidinthe
discovery
anddevelopmentof Seasonal abundance for mosquitoes varies withnatural and chemical pestcontrolstrategies. respect to species. For instance, Culex erythrothorax
In
dairy
operations, cows are washed prior tohas been found to have its peak adult abundance in the
milking,and milking is donetwoorthree times a
day.
summer months in southern California( Seaman 1945), In addition, manure troughs and the milk rooms areand is associated with tule swamps or margins of ponds
washedand cleaned
daily.
Thewaste waterfrom both and rivers where larvae have been collected( Bohart and operationsisremovedfromthebarnviapipes or open Washino 1978). Culex tarsalis Coquillett peaks, inditches to a single or a series of
holding
ponds oradult abundance, from May through November( Reeves
lagoons. Thus, these
dairy
lagoonsare enriched with et al. 1958) and develops in clean rather than pollutedmanure and organic plant material,potentially creating water. Culex quinquefasciatus, the southern house mos-idealconditionsfor Cx.quinquefasciatus andCx.peus quito, has been found in higher numbers from August
development(Mullaet al.1987). These lagoonsrequire
through December and develops in a variety of habitats,
constant surveillance and management(Steelmanet al.including polluted water( Bohart and Washino 1978).
1967, Womeldorfet al.1969,andWhitten1971).
Dairy
Nightly patterns of biting activity of mosquitoes
lagoonswithsomesurrounding vegetationhave been have been documented in California. Anopheles reported to produce
heavy
mosquito populationsfreeborni has peaks of activity near sunset and again
Bailey
etal. 1954, Mullaet al. 1987). Onecultural near sunrise( Cope et al. 1986), whereas, Cx. tarsalispracticeis to
dig deep
well-managed manureholding
had a single peak at sunset( Nelson and Spadoni 1972).ponds and control the resulting vegetation with
Culiseta inornata generally feed at dusk but may
chemicals. Whenthismanagement practiceisrealized,
demonstrate a minor level of feeding behavior during
236 BULL.SOC. VECTOR ECOL. DECEMBER, 1988
the night (Barnard andMulla 1977). Culexerythro-
utilizing CDC traps and human hosts for 10 minute
thoraxhas been noted to seek hostsduring
theday
landing
counts. Relative abundance and population Chapman 1962) in Nevada, andBeck( 1961) found inoscillation in parity rates are presented graphically
Utahthat theyweremostactiveintheevening hours, utilizing a 365
day
calendar (Julian dates).Environ-whereas, in southernCalifornia morning peaks were mental parameters, such as temperature, relative noted
by
Copeetal.( 1986). Peak activityofCx.peushumidity, and wind speed were recorded throughout
has been hard to determine since this species feeds
each night of the study. Studies of the host- seeking
primarilyon passeriformbirds(TempelisandWashino
activity of females in two different physiological states
1967). nulliparous and parous) were taken from subsamples
Studies onthe relationship betweenhost- seeking
from the dry ice surveillance traps at Habitat 3 and
activity and physiological age have been widely
Habitat 5. Parity rates were determined by dissection of
documented. Noapparentrelationship between host- individual females of each species and examination ofseekingandphysiological statewasnotedwithrespect the tracheation of the ovaries( Detinova 1962). toAfricanmosquitospecies(Gilles 1957, Corbet1962), Statistical analysis of the data utilized linear
whileService(1969)noted a positiverelationshipwith
regression techniques for trap catch and prediction
British mosquito species. In California, Nelson and
utility for monitoring population trends ( Sokal and
Spadoni ( 1972) found that no differences in host- Rohlf 1981), Kendall' s coefficient of concordance for seeking activityexisted withrespecttoparitystatefor
peak blood feeding activity for the various time periods
An. freeborni, Aedes vexans ( Meigen), Aedes through the night (Siegel 1956), and the G- test ofmelanimonDyar,andCx. tarsalis.
independence to test parity host- seeking activities
During
thepastseason, theadultmosquitoesina through the night( Sokal and Rohlf 1981).dairy habitat near Norco, California were studied from
JunethroughDecember 1987. The dairies inthis area RESULTS AND DISCUSSION
are surrounded by urban areas and residential housing
which are encroaching upon these semi- agricultural
Mosquito species collected by CDC traps at the
areas. Theobjectivesofthisstudyweretocharacterize
dairy during the 1987 season were predominantly Cx.
adultseasonal populationtrends,spatialdistributionof erythrothorax, Cx. peus, Cx. quinquefasciatus, and Cx. adultsinand around adairy
operation,thephysiological tarsalis. Other mosquitoes found in lower numbersage profileoffemales
during
theentireseason,andthe were An. freeborni and Culiseta particeps. Shifts innighttimeactivitypatternsofhost-seekingadults. abundance of each species varied throughout the
season. However, Cx. quinquefasciatus was the most METHODSANDMATERIALS
prevalent species collected followed by Cx.
erythro-thorax. Culex quinquefasciatus abundance had three The studyareawaslocatedat adairy
1 kmwest of peaks:the first in mid July, the second in early
thecityofNorco in Riverside
County.
Thepredominant September, and the third in late October. The peaks inmosquitoes atthis sitewereproducedmainlyin
dairy
September and October can be explained in part because lagoons,woodland pools, andintailwater.During
thisthe lagoons were reflooded the week before monitoring
study, five distinct habitats were monitored
by
CDCwas initiated, thus, allowing for greater oviposition and
traps: Habitat1, 0. 25kmeastofthe
dairy
lagoonsina larval development. Culex erythrothorax( which does truck crop field; Habitat 2, thedairy/
lagoon proper;not develop in these lagoons) had peaks similar to Cx.
Habitat 3, treerows adjacentto thedairy
andlagoons; quinquefasciatus throughout the season. These peaks Habitat 4, 20meterswestfromthelagoons intreesalong are due to some other yet unidentified environmentalthe interface of the
dairy
operation and an adjacent factors because their development has been reported tohorse/cattle pasture;Habitat5, 0.5 km fromthe
dairy
in be in more permanent bodies of water that support tule ariparianhabitat withbackwaterfrom theSanta Ana growth. Culex peas and Cx. tarsalis had lower numbersriver. Ineachhabitat,twosites were usedtotraphost-
and both had peaks in mid July. Low numbers of Cx.
seeking adult mosquitoes on a semi-monthly basis.
peus in the CO, traps has been reported by Reisen and
Landing
andbiting
activity offemale mosquitoes at Pfuntner( 1987) and similar findings were shown in thisnightwasassessedinalocationnearthe
dairy
lagoonsstudy. All species steadily declined with the onset of
proper. Intheseexperiments, 12all-night studies were
winter, with only Cx. quinquefasciatus adults still active
conducted to determine the activity patterns of adultin the environment( Fig. 1).
mosquitoes. Host- seeking female mosquitoes were Species composition and abundance varied little sampled at various time intervals